The pulmonary nodule “discovered” by pneumonia: a case report
Introduction
The number of patients with pulmonary nodules increased recently because multiple organizations recommend annual low-dose computed tomography (CT) scans for adults with high risk of lung cancer, which has been proved to reduce lung cancer mortality (1). After diagnosed with pulmonary nodules, patients firstly will ask for a definite diagnosis: how likely the nodule is to be benign or malignant? Further, they might ask for proper treatment options: further monitoring, additional biopsy, or surgical removal. We have already got several guidelines for the management pathway for the majority of nodules, including guidelines from British Thoracic Society (BTS) (2) and American Thoracic Society (ATS) (3,4). The recommendations were straightforward with a detailed approach for different types of nodules. We also have several studies that developed composite prediction models for lung nodules (5,6). However, the reality of patients was always more complex. The judgment of clinicians and the compliance of patient might influence the realization of guidelines in clinical practice. Here we report a patient with pulmonary nodules; we try to describe the discovery, monitoring, and treatment of the lung lesions. And we hope this case to provide more clues for the management of such nodules.
Case presentation
A 58-year-old male presented to our hospital on March 14, 2016, because of cough, sputum and left chest pain for 3 days. The symptoms started on March 11, the patient took antibiotics orally (amoxicillin). However, the symptoms did not release. Then he came to our hospital for further diagnosis and therapy. He complained of no dyspnea, fever, chills, or night sweats. The patient had no history of hypertension, diabetes. He had no exposure to tobacco, poisons, or chemicals. The family history was not significant.
On physical examination, he had no crackles or wheezes in the lung. Routine test of blood, urine, and stool were normal. C-reactive protein (CRP) was 25.8 mg/L (normal range was less than 8 mg/L). Carcinoembryonic antigen (CEA) was 1.5 µg/L (normal range was 9.7 µg/L). A chest CT scan on admission showed bilateral bronchopneumonic infiltrates in the upper and lower lobes of both lungs. A solid pulmonary nodule was found in the right upper lobe (Figure 1A-C). The preliminary diagnosis of this patient was “community-acquired pneumonia”. We proposed him to accept active antibiotic treatment (biapenem 0.3 g, iv, 1/8 h, plus levofloxacin 0.5 g, iv, qd).
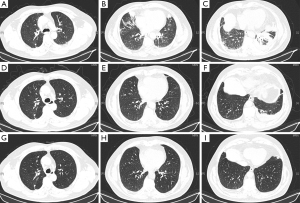
After anti-infectious therapy for 1 week, the symptoms of cough, sputum, and chest pain almost released. On March 22, 2016, the CT scan reported more than 50% inflammation absorption. However, the pulmonary nodule in the right upper lobe was almost the same (8 mm diameter) (Figure 1D-F). We suggested a follow-up of CT scan after 3 months.
On May 3, 2016, the patient came to the outpatient department. He complained he was quite worried about the lung nodule during the past days, and he asked for immediate re-examination. We could find further absorption of inflammation while the pulmonary nodule was still stable. And we considered further follow-up with a longer interval as proper choice for him.
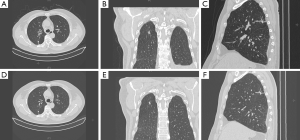
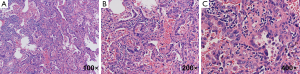
Three months later, the patient came to the specialist clinic with a finished CT scans (Figure 1G-I), he asked for an assured diagnosis of the pulmonary nodule, and the nodule in the right upper lobe infected his quality of life seriously. The serum CEA was 2.1 µg/L then. Three-dimensional reconstruction was performed for the CT scans, and it showed pleural retraction and vascular invasion in the nodule (Figure 2). The patient was admitted to the clinical trial of solitary pulmonary nodules (SPNs) sponsored by Pfizer [The clinical trial to evaluate the value of circulating tumor cells (CTCs) in the differentiation of benign and malignant pulmonary nodules]. We detected the level of CTCs in peripheral blood, and the results were 13 CTCs/3 mL (the cut-off of this research was 8.7 CTCs/3 mL). After detailed discussion with the patient and families, the patient decided to accept a video-assisted thoracic surgery (VATS) approach. Microscopic examination of the nodule reported: high differentiated adenocarcinoma (Figure 3), while immunochemistry (IHC) analysis showed: TTF1 (3+), CK7 (3+), P53 (−), Ki-67 15%. Mutations in the epidermal growth factor receptor (EGFR) tyrosine kinase domain and rearrangements of anaplastic lymphoma kinase (ALK) were not found in this patient. There were no postoperative complications, and the patient was still under follow up till now.
Discussion
There are more and more pulmonary nodules diagnosed with the development of CT technology, and the emphasis of screening for patients with high risk of lung cancer. In fact, most of the diagnosed lung nodules are benign. We have already got various guidelines and prediction models trying to help us to distinguish between benign lesions and malignant nodules. The most mentioned clinical factors included increased age, smoking history, and previous history of extrapulmonary cancer. And the radiologic factors included increased nodule volume, spiculation, upper lobe location, pleural indentation, and volume doubling time less than 400 days (2).
Radiologic analysis
Pulmonary nodules are incidental findings in chest CT scans frequently. They were often divided into solid nodules or sub-solid nodules (SSNs), which were further clarified as part-solid nodules (PSNs) and pure ground glass nodule (PGGNs) (2). Different types of nodules might be included in different management pathways. We further reviewed the prediction models about the pulmonary nodules. The most reported predictors of malignancy in CT scans were: (I) increasing nodule diameter; (II) spiculation; (III) upper lobe location; (IV) pleural indentation (5-7). When coming back to the reported case, the nodule in the right upper lobe was defined as solid nodule about 8 mm diameter. Combined with the clinical characteristics, the patient did not have a history of smoking and extrapulmonary cancer. We offer CT surveillance to this patient.
Then what is the appropriate time interval for this patient? Kostis et al. demonstrated the critical time to follow-up was 12 months for nodules 2–5 mm, 5 months for nodules 5–8 mm, and 3 months for nodules 8–10 mm (8). So, the medical suggestion from us was another CT scans after 3 months initially.
Application of biomarkers
Although several biomarkers, especially circulating markers, have shown interesting early results in differentiating malignant from benign lesions, further researches are required to validate the sensitivity and specificity in defined patients before recommended for clinical application.
Serum CEA, which has been used as a routine screening in our department, were firstly detected for this patient. Chu et al. performed a cross-sectional study, which used CEA, cytokeratin 19 fragments, and neuron-specific enolase for prediction of patients with suspicious pulmonary masses. And the demonstrated sensitivity was only 23.2% (9). It seemed that CEA did not provide sufficient accuracy.
CTCs have been identified to be predictive, prognostic and pharmacodynamic biomarkers in different types of solid tumors, including lung cancer (10,11). Whether the number of CTCs in peripheral blood could be considered as an accuracy biomarker for pulmonary nodules? We look forward to the result of the previously described clinical trial.
Surgical excision
When should the clinician give the suggestion of excision biopsy for the patients? We considered for two situations: (I) we have performed preoperative lung biopsy and got positive results; (II) where a nodule is considered of sufficiently high risk for malignancy (2). For this patient, we made this advisement for the following reasons: (I) although there was not significant volume enlargement of the nodule, the three-dimensional reconstruction of CT scans showed pleural indentation and vascular invasion; (II) the CTCs counts was relatively high in the clinical trial; (III) the patient was quite worried about the nodule, which seriously decreased the quality of life. We need to balance the desire for a certain diagnosis from the patient against the risk of surgery. Researches have demonstrated that patients without preoperative confirmation of malignancy had shorter waiting times, and lower total hospital costs (12). Then the patient was met with another choice: the surgical approach (robotic-assisted lobectomy vs. VATS vs. thoracotomy). Yang et al. found that minimally invasive approaches resulted in similar long-term survival as thoracotomy. Use of VATS resulted, and robotics resulted in the shorter length of hospital stay (13). Our patient finally chose minimally invasive approach of surgery, and there are no symptoms of recurrence till now.
In summary, we described a 58-year-old man with a lung nodule in the right upper lobe, which was occasionally found during a period of pneumonia. We suggested two periods of follow-up, and the patient was also admitted to a clinical trial about CTCs. He finally accepted surgical excision. And the pathologic diagnosis was adenocarcinoma. The clues we might get from this case included: (I) it might be proper for us to offer CT surveillance to patients with solid nodules about 8 mm maximum diameter; (II) when we have a suspicion of malignancy, three-dimensional reconstruction of CT scans could provide more information for the detail of nodules; (III) CTCs counts of peripheral blood showed interest results in preliminary researches, however, further validation was still in needed before clinical practice; (IV) fore nodules with a high risk of malignancy, and did not have the possibility of preoperation biopsy, minimally invasive surgery might be in consideration after fully informed consent.
Acknowledgements
Funding: This work was supported by the Natural Science Foundation of Jiangsu Province of China (BK20140736 to Dong-Mei Yuan), and the Natural Science Foundation of China (81602015 to Dong-Mei Yuan).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395-409. [Crossref] [PubMed]
- Callister ME, Baldwin DR, Akram AR, et al. British Thoracic Society guidelines for the investigation and management of pulmonary nodules. Thorax 2015;70 Suppl 2:ii1-ii54. [Crossref] [PubMed]
- Slatore CG, Horeweg N, Jett JR, et al. An Official American Thoracic Society Research Statement: A Research Framework for Pulmonary Nodule Evaluation and Management. Am J Respir Crit Care Med 2015;192:500-14. [Crossref] [PubMed]
- Wiener RS, Gould MK, Arenberg DA, et al. An official American Thoracic Society/American College of Chest Physicians policy statement: implementation of low-dose computed tomography lung cancer screening programs in clinical practice. Am J Respir Crit Care Med 2015;192:881-91. [Crossref] [PubMed]
- McWilliams A, Tammemagi MC, Mayo JR, et al. Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med 2013;369:910-9. [Crossref] [PubMed]
- Gould MK, Ananth L, Barnett PG, et al. A clinical model to estimate the pretest probability of lung cancer in patients with solitary pulmonary nodules. Chest 2007;131:383-8. [Crossref] [PubMed]
- Swensen SJ, Silverstein MD, Ilstrup DM, et al. The probability of malignancy in solitary pulmonary nodules. Application to small radiologically indeterminate nodules. Arch Intern Med 1997;157:849-55. [Crossref] [PubMed]
- Kostis WJ, Yankelevitz DF, Reeves AP, et al. Small pulmonary nodules: reproducibility of three-dimensional volumetric measurement and estimation of time to follow-up CT. Radiology 2004;231:446-52. [Crossref] [PubMed]
- Chu XY, Hou XB, Song WA, et al. Diagnostic values of SCC, CEA, Cyfra21-1 and NSE for lung cancer in patients with suspicious pulmonary masses: a single center analysis. Cancer Biol Ther 2011;11:995-1000. [Crossref] [PubMed]
- Hou JM, Krebs MG, Lancashire L, et al. Clinical significance and molecular characteristics of circulating tumor cells and circulating tumor microemboli in patients with small-cell lung cancer. J Clin Oncol 2012;30:525-32. [Crossref] [PubMed]
- Krebs MG, Sloane R, Priest L, et al. Evaluation and prognostic significance of circulating tumor cells in patients with non-small-cell lung cancer. J Clin Oncol 2011;29:1556-63. [Crossref] [PubMed]
- Sihoe AD, Hiranandani R, Wong H, et al. Operating on a suspicious lung mass without a preoperative tissue diagnosis: pros and cons. Eur J Cardiothorac Surg 2013;44:231-7; discussion 237. [Crossref] [PubMed]
- Yang HX, Woo KM, Sima CS, et al. Long-term Survival Based on the Surgical Approach to Lobectomy For Clinical Stage I Nonsmall Cell Lung Cancer: Comparison of Robotic, Video-assisted Thoracic Surgery, and Thoracotomy Lobectomy. Ann Surg 2017;265:431-7. [Crossref] [PubMed]


