
IgA vasculitis and polymyalgia rheumatica induced by durvalumab
A 64-year-old man, former smoker with no prior relevant medical history was diagnosed with squamous cell carcinoma of the lung and underwent right bilobectomy (MRL and LRL) and mediastinal lymphadenectomy. The histopathology report confirmed the tumor to be pT1aN1M0. The patient was offered adjuvant chemotherapy with cisplatin and vinorelbine followed by adjuvant treatment with durvalumab (anti-PD-L1) 20 mg/kg/28 d in a clinical trial (NCT02273375). After 5 cycles of durvalumab, he presented at the emergency department with a 48-hour history of inflammatory pain in shoulders and pelvis, as well as low-grade fever. Physical examination revealed functional limitation of scapular and pelvic girdle. Laboratory analysis revealed increased acute-phase reactants (CRP 52 mg/L; ESR 92 mm/h). Ultrasound of the shoulders showed bilateral bicipital tenosynovitis and subacromial bursitis, thus confirming the diagnosis of polymyalgia rheumatica (PMR), which was associated with durvalumab. Treatment with methylprednisolone 12 mg/d led to a good clinical and laboratory response; therefore, immunotherapy was maintained. Three weeks later, while the patient was undergoing methylprednisolone 8 mg/d, he presented with erythematous macules and papules and edema on the lower limbs and was admitted for further assessment. Skin biopsy revealed leukocytoclastic vasculitis. The results of an immunological study based on antinuclear antibodies, antineutrophil cytoplasmic antibodies, rheumatoid factor and cryoglobulins were negative. Complement levels were normal. Twenty-four-hour urine test revealed proteinuria (539 mg/24 h) and hematuria (90% dysmorphic red blood cells). PET-CT ruled out recurrence of the lung tumor. Kidney biopsy eventually revealed IgA vasculitis. In summary, we present the case of a patient receiving durvalumab who presented with symptoms compatible with PMR and IgA vasculitis. Given the second immune related adverse event (IR-AE) induced by durvalumab, we decided to withdraw immunotherapy and start treatment with angiotensin converting enzyme inhibitors and 0.5 mg/kg/d prednisone. The symptoms were completely resolved and it was possible to taper glucocorticoids dose till its withdrawal 1 year later. The patient is currently being followed at the oncology and rheumatology clinics, is not receiving cancer treatment nor rheumatic, and remains symptom-free.
The advent of the immune check-point inhibitors (ICI) in the clinical scenario has meant a relevant change in the therapeutic approach for several solid tumors. They have changed dramatically the prognosis of these tumors with a substantial improvement of survival and even with long-lasting responders to such therapies. Despite this, ICI have come together with a new spectrum of toxicities. Generally, the incidence of the IR-AE is low with a mild or moderate symptomatic burden at presentation. Usually, they can be well controlled with steroids, requiring dose delays and occasionally drug withhold. Occasionally, these IR-AE may be life-threatening with permanent and disabling sequels (1).
Here we describe a case of PMR with associated IgA vasculitis secondary to durvalumab administration. We hypothesized that the dysregulation of the immune system secondary to ICI action led to the development of different autoimmune processes in the same patient, which usually don’t appear simultaneously in clinical practice. Moreover, it is interesting to know that the development of an IR-AE (rheumatic or not) may increase the possibility to develop a second IR-AE (1-4).
PMR has been previously described as a rheumatologic IR-AE, but vasculitis is uncommon, with few cases reported in the literature (5). To date, there have been 24 cases of vasculitis associated with ipilimumab (n=8), nivolumab (n=6), pembrolizumab (n=7), and combination therapy (n=3), with the most frequent being large vessel vasculitis (n=8), central nervous system and peripheral primary vasculitis (n=7), followed by small vessel vasculitis (n=6), and, finally, a miscellaneous group that does not fit with those described above (n=3). These data are summarized in Table 1. To the best of our knowledge, here we present the first case of IgA vasculitis induced by an ICI reported in the literature.
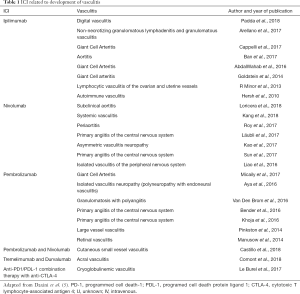
Full table
In conclusion, given the growing use of ICI in cancer, the frequency of IR-AE is likely to increase. Therefore, the awareness of their occurrence and a multidisciplinary approach for an early diagnosis and management are crucial to guarantee the treatment adherence and minimize the impact in patients’ quality of life.
Acknowledgments
The authors thank Sociedad Española de Reumatología for assistance in drafting the manuscript.
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tlcr.2020.03.01). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Postow MA, Sidlow R, Hellmann MD. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N Engl J Med 2018;378:158-68. [PubMed]
- Cappelli LC, Gutierrez AK, Bingham CO, et al. Rheumatic and Musculoskeletal Immune - Related Adverse Events Due to Immune Checkpoint Inhibitors: A Systematic Review of the Literature. Arthritis Care Res (Hoboken) 2017;69:1751-63. [Crossref] [PubMed]
- Kostine M, Rouxel L, Barnetche T, et al. Rheumatic disorders associated with immune checkpoint inhibitors in patients with cancer—clinical aspects and relationship with tumour response: a single-centre prospective cohort study. Ann Rheum Dis 2018;77:393-8. [Crossref] [PubMed]
- Cappelli LC, Shah AA, Bingham CO. Immune-related adverse effects of cancer immunotherapy – Implications for rheumatology. Rheum Dis Clin North Am 2017;43:65-78. [Crossref] [PubMed]
- Daxini A, Cronin K, Sreih AG. Vasculitis associated with immune checkpoint inhibitors-a systematic review. Clin Rheumatol 2018;37:2579-84. [Crossref] [PubMed]


