
Immuno-based therapeutic strategies for initial unresectable locally advanced non-small cell lung cancer: a case report
Introduction
Globally, lung cancer is responsible for more deaths than any other cancer. The clinical management of locally advanced disease, which accounts for about 30% of non-small cell lung cancer (NSCLC) cases, is highly challenging because of the poor prognosis, even in the wake of multidimensional treatment modalities (1).
In recent years, therapeutic strategies for patients with NSCLC and many other tumor types have been changed enormously by immunotherapy based on immune checkpoint inhibitors. Despite the encouraging outcomes of neoadjuvant immunotherapy seen in recent trials, such as the NEOMUN and NADIM trials, the prognoses of patients with initially unresectable NSCLC with T4 or bulky swollen N2 lymph nodes are still unsatisfying (2,3). For this reason, novel therapeutic modalities are desperately needed. Here, we present our experience of an immuno-based therapeutic strategy for a patient with initially unresectable NSCLC with T4 and bulky swollen N2 lymph nodes. We present the following case in accordance with the CARE reporting checklist (available at http://dx.doi.org/10.21037/tlcr-20-517).
Case presentation
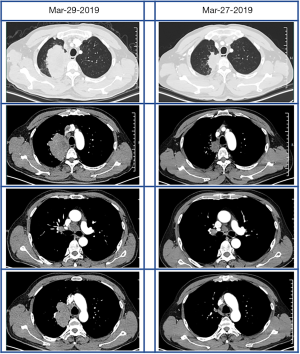
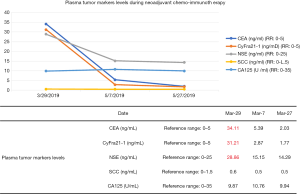
A 56-year-old male with no history of smoking was admitted to the hospital complaining of non-productive cough on March 29, 2019 (Figure 1). A chest CT scan revealed a heterogeneous, infiltrative, 7.7 cm × 6.2 cm mass in the patient’s right upper lung with bulky swollen mediastinal and hilar lymph nodes (Figure 2). Percutaneous CT-guided needle biopsy revealed adenocarcinoma from the mass. 18F-FDG positron emission tomography (PET) indicated positive mediastinal and hilar lymph nodes (stations 2, 4, 7, and 10). Molecular testing illustrated pan-negative results for commonly known oncogenic driver mutations. Ventana ALK (D5F3, Ventana Medical Systems Inc., Oro Valley, AZ, USA) immunohistochemistry (IHC) also demonstrated a negative result. IHC test of CST-PD-L1 (E1L3N®) detected a PD-L1-positive rate of 10% in the NSCLC tumor cells. Consequently, the patient was diagnosed with stage IIIB (T4N2M0) lung adenocarcinoma. After discussing the patient’s case, the lung cancer multi-disciplinary team (MDT) decided on a treatment of pemetrexed and carboplatin, in combination with pembrolizumab 200 mg every 3 weeks. After 2 cycles of chemo-immunotherapy, a repeat chest CT scan revealed that the right upper lung mass had reduced in size to 4.6 cm, with shrinking mediastinal and hilar lymph nodes (Figure 2). A steady decrease in the levels of plasma tumor marker, such as CEA, CyFra21-1, and NSE, was also observed (Figure 3).
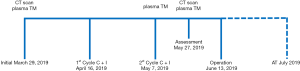
Another lung cancer MDT discussion after the second cycle of chemo-immunotherapy concluded that the patient would undergo surgical treatment. Four weeks after the completion of the neoadjuvant chemo-immunotherapy course, a right upper lobectomy with mediastinal lymphadenectomy was performed. The final pathology report revealed a 5.5-cm, T3N2 adenocarcinoma. The patient’s postoperative courses include subsequent adjuvant immunotherapy for a one-year period, as the adjuvant treatment in the NADIM trial, and radiotherapy.
Discussion
Locally advanced NSCLC represents a heterogeneous group of tumors, varying from resectable types with microscopic lymph nodes metastases to unresectable tumors with multiple bulky swollen N2 lymph nodes. During recent decades, the benefit of neoadjuvant chemotherapy for the survival rate of such patients has been put forward (4). The majority patients with locally advanced NSCLC whose disease is initially unresectable experience disease progression, despite definitive concurrent chemoradiotherapy (5). For the patient in our case report, despite being defined as initially unresectable, he was judged as a potentially resectablecase following the MDT discussion, based on Chinese guidelines for diagnosis and treatment of primary lung cancer (6). Therefore, neoadjuvant therapy, rather than definitive chemoradiation, was recommended to this patient.
Previous studies have demonstrated that elevated levels of PD-L1+ lung cancer cells and tumor-associated immune cells after neoadjuvant chemotherapy generate a favorable response to immunotherapy (7). These results encourage the combination of chemotherapy and immunotherapy in neoadjuvant therapy for locally advanced NSCLC.
The timing of additional adjuvant immunotherapy presents a couple of interesting questions. Firstly, the best strategy for delivering additional adjuvant immunotherapy remains unclear. Based on limited preliminary clinical evidence, this patient was recommended to undergo additional adjuvant immunotherapy. Secondly, liquid biopsy of immunological markers, such as T cell clonal dynamics and CD8+ T cell subsets, should be a focus of exploratory research (8,9).
Conclusions
Our case study suggests that immuno-based therapeutic strategies present alternative options for patients with initially unresectable locally advanced NSCLC with T4 or bulky swollen N2 lymph nodes.
Acknowledgments
Funding: This work was supported by the National Natural Science Foundation of China (No. 81702251 and 81972176), the National Natural Science Foundation of Shanghai (No.18ZR1435100) and Shanghai Hospital Development Center (SHDC12016113).
Footnote
Reporting Checklist: The authors have completed the CARE reporting checklist. Available at: http://dx.doi.org/10.21037/tlcr-20-517
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/tlcr-20-517). The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Karachaliou N, Fernandez-Bruno M, Bracht JW, et al. Challenges and unanswered questions for the next decade of immune-oncology research in NSCLC. Transl Lung Cancer Res 2018;7:691-702. [Crossref] [PubMed]
- Borghaei H, Langer CJ, Gadgeel S, et al. 24-Month Overall Survival from KEYNOTE-021 Cohort G: Pemetrexed and Carboplatin with or without Pembrolizumab as First-Line Therapy for Advanced Nonsquamous Non-Small Cell Lung Cancer. J Thorac Oncol 2019;14:124-9. [Crossref] [PubMed]
- Eichhorn F, Klotz LV, Bischoff H, et al. Neoadjuvant anti-programmed Death-1 immunotherapy by Pembrolizumab in resectable nodal positive stage II/IIIa non-small-cell lung cancer (NSCLC): the NEOMUN trial. BMC Cancer 2019;19:413. [Crossref] [PubMed]
- Rosenzweig KE, Gomez JE. Concurrent Chemotherapy and Radiation Therapy for Inoperable Locally Advanced Non-Small-Cell Lung Cancer. J Clin Oncol 2017;35:6-10. [Crossref] [PubMed]
- Bradley JD, Paulus R, Komaki R, et al. Standard-dose versus high-dose conformal radiotherapy with concurrent and consolidation carboplatin plus paclitaxel with or without cetuximab for patients with stage IIIA or IIIB non-small-cell lung cancer (RTOG 0617): a randomised, two-by-two factorial phase 3 study. Lancet Oncol 2015;16:187-99. [Crossref] [PubMed]
- Chinese guidelines for diagnosis and treatment of primary lung cancer 2018 (English version). Chin J Cancer Res 2019;31:1-28. [Crossref] [PubMed]
- Parra ER, Villalobos P, Behrens C, et al. Effect of neoadjuvant chemotherapy on the immune microenvironment in non-small cell lung carcinomas as determined by multiplex immunofluorescence and image analysis approaches. J Immunother Cancer 2018;6:48. [Crossref] [PubMed]
- Liu J, O'Donnell JS, Yan J, et al. Timing of neoadjuvant immunotherapy in relation to surgery is crucial for outcome. Oncoimmunology 2019;8:e1581530. [Crossref] [PubMed]
- Zhang J, Ji Z, Caushi JX, et al. Compartmental Analysis of T-cell Clonal Dynamics as a Function of Pathologic Response to Neoadjuvant PD-1 Blockade in Resectable Non-Small Cell Lung Cancer. Clin Cancer Res 2020;26:1327-37. [Crossref] [PubMed]


