MET inhibition in lung cancer
Introduction
The discovery of new cancer-driver genes and the enforcement of molecules targeting them have changed the landscape of Non Small Cell Lung Cancer (NSCLC) treatment.
As a matter of fact, the previous scenario of advanced NSCLC treatment has been completely revolutionized, switching from a “one size fits all” approach to a personalized therapy.
Somatic mutations of the Epidermal Growth Factor Receptor (EGFR) tyrosine kinase domain positively correlated with clinical responsiveness to specific inhibitors: gefitinib, erlotinib and afatinib, two reversible and one irreversible EGFR inhibitors, have consistently demonstrated significant increase of Response Rate (RR) and Progression-Free-Survival (PFS) compared to standard chemotherapy in EGFR mutated NSCLC patients with advanced disease (1-7).
The Anaplastic Lymphoma Kinase (ALK), firstly identified from a chromosomal translocation leading to the production of merged proteins in Non-Hodgkin lymphomas, was then detected as a fusion with the echinoderm microtubule-associated protein-like 4 (EML4) in 6.7% of NSCLC patients (8,9). Crizotinib (PF02341066, Xalkori) targets EML4-ALK thus gaining astonishing response rates in a phase I/II trial and more recently in a phase III trial (10,11).
Unfortunately, other biomarkers already identified in NSCLC are still “undraggable” and one clear example is KRAS. KRAS is a member of the RAS family of oncogenesis, involved in signal transduction and tumorigenesis and its mutations, frequently in codons 12 and 13, have been reported in 20-30% NSCLCS (12-15). Some sign of activity came in the last year from a targeted agent (Selumetinib), which compared to standard chemotherapy in KRAS mutated patients gave interesting results in terms of RR and PFS (16).
Several other molecular markers’ alterations have been described in NSCLC such as: phosphatidylinositol 3-kinases (PI3K) (2%), lipid kinases that regenerate a key mediator between growth-factor receptors and intracellular downstream signaling pathways; ERBB-2 (2%); B-RAF (1-3%), a Ser-Thr kinase that links RAS GTPases to downstream proteins of the MAPK family, thus controlling cell proliferation; ROS1 (about 1%), oncogene that encodes a transmembrane tyrosine kinase receptor; AKT; RET and MET (17-21).
Since the first MET pathway description, several inhibitors have been preclinically and clinically tested, both alone and in combination with chemotherapy or other targeted therapies.
This paper will focus on MET biology, its role in the cell function and tumorigenesis, specifically in lung cancer, as well as on the molecules that target it.
Met discovery and mechanism of action
Met is a heterodimer receptor tyrosine kinase composed of a α-chain and a β-chain, linked by a disulphide bond.
Met was originally isolated as the product of a human oncogene, trp-met, in tumor cells treated with a chemical carcinogen. Met gene encodes a 170-kD protein (p170met) that has constitutive and ligand-independent tyrosin-kinase activity. Met has pivotal functions in embryogenesis and organogenesis of placenta, liver, kidney, neurons and muscles (22-25).
Moreover, in vivo, Met receptor activation determines a phenomenon called “invasive growth”, which includes cell proliferation, scattering, survival, motility and invasion, epithelial-mesenchymal transition and branched morphogenesis (26,27).
The natural ligand for this receptor is the HGF, produced by stromal and mesenchymal cells, that acts primarily on Met-expressing epithelial cells in an endocrine and/or paracrine fashion (24,28). HGF-induced Met tyrosine kinase activation is regulated by paracrine ligand delivery, ligand activation at the target cell surface and ligand-activated receptor internalization and degradation (29). Going more into details, when HGF binds to the Met receptor, Met major autophosphorylation sites (located within the tyrosine kinase domain) are phosphorylated, with subsequent intrinsic catalytic activation of multiple signaling cascades involved in cell proliferation, survival, angiogenesis, morphogenesis, cell scattering, motility, migration and invasion. An activated docking site in the kinase domain further recruits intracellular adaptor molecules through the SH2 domains and other recognition motifs, such as GAB1 (a key coordinator of the cellular responses to Met). Downstream signaling of the GRB2-mitogen-activated protein kinase (MAPK) cascade, PI3K-mTOR pathway, and STAT pathway are eventually activated, mediating various cellular functions (27,30,31). Finally, in order to activate the receptor, proteolytic cleavage of proHGF is necessary (25).
HGF is mainly produced by stromal tissue like liver and bone marrow, and is expressed in a multitude of mesenchymal-derived cells. Being Met expression detected in the epithelium of most tissues, this indicates that HGF-Met signal transduction pathway contributes to mesenchymal-epithelial interactions (24,32-34).
Met downregulation occurs through rapid internalization of Met itself and subsequent degradation by the lysosome: this process is regulated by ligand-dependent ubiquitination of Met, a process also modulated by specific tyrosine phosphatases and recently identified as proteins decorin and LRIG1 (35,36).
Met can be altered through receptor overexpression, genomic amplification, mutations or alternative splicing. These alterations lead to signaling deregulation that can be mediated through ligand (HGF)-independent receptor activation or through its ligand (HGF)-dependent activation via autocrine (intratumoral HGF), paracrine (mesenchymal or microenvironmental HGF), or endocrine (circulatory HGF) loop signaling cascades (29).
HGF and Met are highly expressed in various stem and progenitors cells, but are only expressed as low levels in their mature cells (25). In preclinical animal models, whereas the overexpression of Met and/or HGF has been shown to stimulate tumorigenesis and metastasis, down-regulation of Met or HGF expression resulted in increased apoptosis and decreased tumor growth and blood vessel density (37-40). Moreover, Met interacts synergistically with VEGF to promote angiogenesis, cell proliferation and invasion (41). This occurs through the transcriptional up-regulation of the hypoxia inducible factor-1α and amplified HGF signaling, that resulted in both induction of invasion and increased expression of VEGF (41).
Met pathway is also one of the key players in the development of acquired resistance to VEGF pathway inhibitors: the inhibition of Met expression prevented hypoxia-induced invasion growth (42,43).
The increased Met expression described in case of response to ionizing radiation through the ATM-NFκB signaling pathway, could lead to radioresistance and cancer invasion (44).
Met pathway and cross-talks
The cross-talk of Met with various signaling pathways is described in literature and that one between Met and EGFR/HER family receptors is particularly important in lung cancer (45-49).
Met and EGF family receptors are often described co-expressed in tumors and transactivation of Met depends on elevated expression of EGFR in many human tumors (46,50,51). Conversely, HGF stimulation promotes transactivation of EGFR in multiple cell lines, including NSCLC (49).
Cooperation between Met and EGFR occurs also indirectly: when Met activates Src, this lead to EGFR phosphorylation and the creation of docking sites for EGFR interactors involved in downstream signaling (52).
Moreover, through receptor cross-talk, Met exerts a key role in the development of resistance to EGFR family inhibitors. One example is the stimulation of HER-3 phosphorylation and signaling to Akt (a key signaling molecule required for cell survival and proliferation) when Met is amplified and overexpressed (53,54). Inhibition of Met in EGFR inhibitors resistant cells, either in vitro or in vivo, promotes apoptosis, tumor growth reduction and significant necrosis (49,53).
Met and EGFR inhibitors combined together, cooperatively abrogate ErbB3 signaling activation (49). An alternative mechanism in this context is the Src-induced EGFR phosphorylation (52).
Preclinical data also support that Met cross-talks and cooperates with other members of the EGF receptor family, including HER2, to enhance cell invasion and this lead to the possibility to explore therapeutic activity of dual Met and HER2 therapies (55,56).
Stimulation with both HGF and EGF enhances downstream activation of several signaling pathways including Akt, Erk and STAT3 in a way that Met inhibitors abolished their baseline phosphorylation (57,58).
The already mentioned interaction between decorin and LRIG1 proteins, promotes ligand-independent receptor downregulation and degradation of EGFR family members. Decorin binds to the EGFR family, inducing receptor dimerization, internalization and eventual lysosomal degradation, whereas LRIG1 and EGFR associated via their extracellular domains, allow enhanced EGFR phosphorylation. Thus, Met promotes resistance to VEGFR and EGFR inhibitors (59,60).
Cross-talk between Met and KRAS signaling has also been described both in preclinical and clinical findings (61,62). Met activates RAS directly or via a protein-tyrosine phosphatase (63). Similarly, PI3K could be directly activated by Met or indirectly by RAS protein (30).
Moreover, Met directly binds to and sequesters the Fas receptor. This interaction prevents Fas self-aggregation and ligand binding, thus inhibiting Fas activation and apoptosis (64).
Finally, preclinical studies exploring a combination of anti-Met therapeutic agents with mTOR inhibitors have also demonstrated an increased growth suppression, compared to mTOR inhibitors alone (62).
Met plays also a functional role in signaling pathways mediated by other membrane proteins. Integrin-dependent signaling could trigger ligand-independent Met phosphorylation following cellular adhesion, and Met and integrins might have independent yet synergistic roles in cell invasion. Plexins, single-pass transmembrane receptors for semaphorins, acts cooperatively with Met for cell adhesion and migration (45).
MET and NSCLC
Met receptor is overexpressed in both Small Cell Lung Cancer (SCLC) and NSCLC, mainly in non-squamous histotype (65-67).
Recent tumor microarray expression analysis demonstrated a 72% Met expression in human lung cancer tissue and 40% Met receptor over-expression; such values are higher than in breast (16%) and ovarian cancer (31%), but lower than in renal (70%) and colorectal cancers (CRC; 78%) (67). Phospho-Met expression is found to be at the highest levels in lung cancer (73%), followed by ovarian (33%), breast (23%), and renal (18%) cancer (67).
Met gene amplification can guide the dependency of cell survival and proliferation upon the Met signaling, even in lung cancer cell lines. Blocking Met causes significant growth inhibition, G1-S arrest and apoptosis in cell lines harboring Met gene amplification. When Met is not amplified, its levels of activation are low and cells are unable to grow (68).
Different studies have reported primary Met amplification to be in the wide range of 2% to 21%, in NSCLC lung adenocarcinomas, particularly in TKI-naïve cohorts (69-72).
In lung cancer, Met receptor mutations were mainly found clustered in the non-tyrosine kinase domain, in the juxtamembrane (JM) domain and in the sema domain (67). These mutations are oncogenic activating variants, that result in a deletion in the juxtamembrane domain with enhanced oncogenic signaling, tumorigenicity, cell motility, and migration (27,73). Met kinase domain mutations have been found to be somatically selected in the metastatic tissues, compared with the primary solid cancers (74).
Literature data are quite discordant on the prognostic value of Met over-expression, amplification and mutation.
The overexpression of circulating Met in patients with NSCLC has been strongly associated with early tumor recurrence and patients with adenocarcinoma and Met amplification have also demonstrated a trend for poor prognosis (69,75,76).
Concerning the correlation between Met FISH status and clinical characteristics, only Okuda and colleagues demonstrated an association with male gender and smoking status, showing also a relationship with high Met gene copy number (77). In the same trial, both FISH positive and gene amplified cases had a worse prognosis, although the difference was not statistically significant and among the Met FISH-positive NSCLCs, patients with gene amplification showed not significantly worse OS compared to those with high polysomy.
All FISH-positive cases had squamous histology, adenocarcinoma had Met amplification: high Met gene copy number tended to have shorter OS and PFS than those with low Met gene copy number, being this difference statistically significant only in the squamous histotype.
Moreover, at multivariate analysis done on squamous histology, increased Met gene copy number and Met amplification were confirmed to be independent poor prognostic factors.
No significant difference in prognosis was found in patients having adenocarcinoma regardless Met FISH status in the korean study. In contrast, Beau-Faller and colleagues found a tendency toward shorter event-free survival in adenocarcinoma patients with increased Met gene copy number, whereas Kanteti and colleagues demonstrated that the high Met gene copy number in adenocarcinoma was associated with a trend of better prognosis (69). However, the above mentioned study has some critical methodology aspects as it was conducted on a small sample size and qPCR was used as test and not FISH, done on DNA samples extracted from formalin-fixed paraffin-embedded (FFPE) archival tumor tissues (70).
Capuzzo and colleagues found no patient with EGFR mutation was Met FISH positive, but increased Met gene copy number significantly correlated with EGFR FISH-positive status (78).
Acquired Met amplification has also been linked to approximately 22% of non-T790M mediated secondary gefitinib resistance in NSCLC patients, although it can also occur concurrently but independently (52,53,78-80).
Using in vitro cell line models, the Met gene amplification in gefitinib-resistant cell clones was identified (53).
Rho and colleagues tried to demonstrate that Met activation, rather than gene amplification, is sufficient to promote EGFR resistance, but the activation appear to be secondary to increase passage numbers rather than to EGFR-Tki exposure (81).
More recently, two prospective analyses have investigated the mechanism of EGFR-Tki resistance through the tissue rebiopsy: high Met gene copy number was found in 11% and 5% of the tissue samples, respectively (82,83).
Met inhibitors
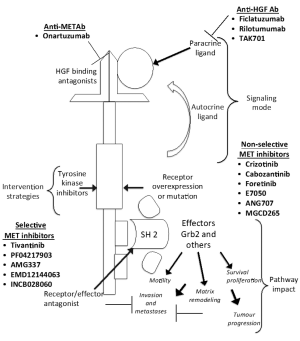
Several inhibitors have been tested so far: they can be classified according to their mechanism of action in selective Met inhibitors, unselective Met inhibitors and antibodies targeting Met or HGF (Figure 1, Table 1).
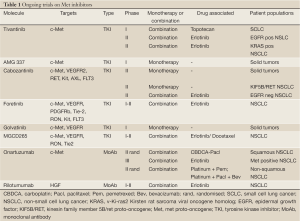
Full table
Selective Met inhibitors
Tivantinib
Tivantinib (ARQ 197) is the first non-ATP-competitive small molecule that selectively targets the Met RTK, locking and stabilizing the kinase in a “closed” and “inactive” conformation, causing the disruption of Met phosphorylation and the downstream signaling.
Moreover, tivantinib enhances Met degradation through the ubiquitin/proteasome pathway in vitro, induces apoptosis in Met activated cell-lines and it’s active in multiple human cancer xenografts (84,85).
Tivantinib acts synergically with antiangiogenentc drugs in preclinical studies on solid tumor cell lines (86).
Studies in vitro and in vivo demonstrated its activity in several types of cancer such as breast, colorectal and gastric cancer (85,87).
Met cancer expressing cell lines treated with tivantinib displayed either a dose-dependent loss of proliferative capacity or caspase-dependent Met apoptosis, which positively correlated with either ligand-dependent Met activity or constitutively active Met. Tivantinib does not exert any activity in cancer cell lines not expressing Met or phospho-Met.
Tivantinib has been investigated in three phase I trials, as single agent and in combination.
In the first dose-escalation phase I trial, tivantinib is administered as single-agent in patients with advanced solid tumors. Initially, an intermittent dosing was planned but, due to the bradycardia experienced in the other phase I trial using this schedule, the protocol was amended and the following 79 patients received a continuous dose (88).
No MTD was reached in this study and less than 33% of patients experienced DLTs at any given dose. Thus, the recommended phase II dose was confirmed at 360 mg twice a day as per a concomitant phase I study, where this MTD was identified (88).
The most commonly reported drug-related adverse events of any grade included fatigue, gastrointestinal (GI) disorders (nausea, vomiting and diarrhea) and anemia.
Pharmacokinetic was linear. There was considerable inter-patient variability, but no relationship between drug-related adverse events (AEs), dose and extent of tivantinib exposure; consequently, this inter-patient variability was not considered relevant for its clinical safety. Partial responses registered in this trial were equal to 4.8% (89).
In another phase I trial, two formulations of tivantinib were tested: the amorphous and the crystalline A formulation. The trial was lead in a single institution, the Royal Marsden Hospital (Sutton, United Kingdom) and highlighted the following DLTs: one patient had grade 3 fatigue at 200 mg, one patient presented a grade 3 febrile neutropenia, one other a grade 3 mucositis, one a grade 3 palmar-plantar erythrodysesthesia and one a grade 3 hypokalemia at 400 mg. The MTDs- recommended phase 2 doses (RP2Ds) were 300 mg bi-daily for the amorphous formulation and 360 mg bi-daily for the crystalline A formulation. The main grade 1-2 AEs, all generally self-limiting, were fatigue (15.7%), nausea (13.7%), vomiting (11.8%). Tivantinib is metabolized by CYP2C19: one patient with CYP2C19 deficiency experienced grade 4 febrile neutropenia and grade 3 mucositis as the drug’s AUC was 3-fold higher (90).
The crystalline A formulation of tivantinib resulted in lower drug exposure at 300 and 360 mg twice daily, compared with the amorphous form at 300 mg twice daily (likely due to different dissolution characteristics). RECIST stable disease ≥4 months was the best response in 14 patients, together with minor tumor regressions (88).
As the ratio of the poor metabolizers of CYP2C19 in Asians is around 20% (while is very low in Caucasians), a Japanese phase I trial was designed to evaluate drug’s safety profile of tivantinib in this group of patients with metastatic solid tumors and the drug was well tolerated, but CYP2C19 genotype clearly affected the exposure and the RP2Ds differed for “no poor metabolizers” (360 mg bi-daily) and for “poor metabolizers” (240 mg bi-daily). Most common AEs were similar to those mentioned above (91). A phase III trial was conducted in Asia in advanced NSCLC patients, comparing erlotinib + tivantinib versus erlotinib + placebo at the dose calculated considering the CYP2C19 polymorphism (92). A press release in August 2012 announced a suspension in the accrual for this study, due to suspected cases of interstitial lung disease (93).
Based on the preclinical data showing a synergistic action between EGFR-TKi and Met inhibitors, an open-label sequential dose escalation phase I trial on tivantinib + erlotinib was set up. Thirty-two metastatic cancer patients were included: 59% were males, 75% PS 1 and mean age was 60 years. The MTD was not established, however, the RP2D was 360 mg bi-daily for tivantinib and 150 mg daily for erlotinib. Two DLT were experienced at 360 mg (grade 4 neutropenia, grade 3 thrombocytopenia), none at 240 or 120 mg. The most common AEs were cutaneous rash, fatigue, nausea, abdominal pain, diarrhea, bradycardia and anemia, mostly grade 1 and 2. No drug related death, but 11% grade 3-4 neutropenia and 8% grade 3-4 nausea were recorded (94).
This combination of erlotinib (150 mg daily) + tivantinib (360 mg bi-daily) every 4 weeks was further studied in a phase II, double-blind, randomized open-label study in comparison with erlotinib 150 mg daily + placebo, in previously treated locally advanced or metastatic NSCLC patients. One hundred and sixty-seven patients were enrolled and homogeneously distributed between the two arms (mainly males, never or former smoker, with stage IV disease and adenocarcinoma histology): 10% in the combination arm versus 18% in the standard arm presented an EGFR mutation, 10% versus 17% a KRAS mutation, 26% versus 26.5% had 4 or more MET gene copy number. The ORR was 10% for erlotinib + tivantinib versus 7% for the control arm.
Median investigator’s PFS was 3.8 months for the tivantinib + erlotinib arm versus 2.3 months for the erlotinib + placebo arm (HR=0.81, P=0.24); the reviewer’s PFS was 3.6 versus 2 months (HR=0.74, P=0.09). Median OS was 8.5 for the investigational arm versus 6.9 months for the control arm (HR=0.87, P=0.47). Pre-planned exploratory survival analysis in non-squamous histology showed a trend of benefit from the combination arm in both PFS (HR=0.71) and OS (HR=0.72). Even in a small number of patients, the subgroup analysis showed an advantage in terms of PFS for EGFR wild type (HR=0.70), KRAS mutated patients (HR=0.76) and for Met FISH positive patients (>5, HR=0.45).
Treatment was well tolerated both in the investigational and in the control arm: low grade rash (9.5% versus 7.2%) and diarrhea (7.1% versus 7.2%), fatigue (4.8% versus 6%), nausea (1.2% versus 4.8%), vomiting (3.6% versus 1.2%), dyspnea (7.1% versus 13.3%), anemia (6% versus 7.2%) were the most common reported toxicities (95).
On the basis of data coming from this phase II trial, the phase III MARQUEE trial was designed in non-squamous NSCLC patients with the same schema, having the overall survival (OS) as primary end-point. Unfortunately, a press release in October 2012, revealed that the primary end point in the intent to treat population was not met, but no further data are yet available (96,97).
Others selective Met inhibitors
PF-04217903 is a selective ATP-competitive small inhibitor of Met kinase. It inhibits tumor cell proliferation, survival, migration/invasion in Met-amplified cell lines in vitro, and shows marked antitumor activity in tumor models harbouring either Met gene amplification or a HGF/Met autocrine loop. PF-04217903 also demonstrates potent antiangiogenic properties in vitro and in vivo (98). In 2012 a phase I trial with PF-04217903 in patients with advanced solid tumors was prematurely discontinued, due to strategic development decision by Pfizer. No safety concerns were reported (99).
AMG 337 is a selective inhibitor of the proto-oncogene Met thereby disrupting Met signal transduction pathway. A phase I, open-label, sequential dose escalation and expansion study with AMG 337 in subjects with advanced solid tumors is currently ongoing (100) (Table 1).
INCB028060 is an oral potent and highly selective Met inhibitor, capable of suppressing tumor growth in vivo at doses that are extremely well tolerated (101,102).
Good tolerance was confirmed in a phase I standard 3+3 dose-escalation study once or twice daily on a continuous 28-day schedule in patients with advanced solid tumors. The MTD was not reached and no grade 3-4 AEs were noted, except grade 3 ALT increase in a patient with liver metastases and grade 2 ALT levels at baseline. Grade 1-2 AEs experienced were mild tremor, fatigue, nausea, diarrhea, indigestion and headache (103).
Non-selective Met inhibitors
Crizotinib
Crizotinib was synthesized primarily as a Met inhibitor. It was engineered based on interactions of a precursor (PHA-665752) with the ATP-binding sites of the Met kinase domain thus resulting in displacement of the kinase activation loop, that interferes with ATP and substrate binding to the Met receptor tyrosine kinase. Crizotinib was designed in order to be less lipophilic and to have a small hinge binder with the possibility to better interact in the kinase pocket (104).
Crizotinib was proved to be active in NSCLC cell lines carrying Met amplification. However, no activity was described in Met mutated, EGFR mutated or normal cell lines. Moreover, crizotinib markedly inhibited AKT, Met and ERK phosphorylation. By doing that, it induced apoptosis even though a mediation of BIM up-regulation (pro-apoptotic member of the Bcl-2 family) and survivin down-regulation (a member of the inhibitor of apoptosis protein family) has also been reported.
Interestingly, in Met or EGFR mutated but also in normal cell lines, whit a low Met phosphorylation, the Met phosphorylation is completely inhibited, whereas the ERK and AKT are not (105).
During drug development, Ou and colleagues described a case of prolonged partial response to crizotinib in a NSCLC patient carrying Met amplification (defined as Met/CEP7 ratio >5) but no ALK translocation (106).
The first phase I trial was designed as open-label, multicenter, to evaluate safety and efficacy of crizotinib: this study was emended with an expanded cohort for patients with lung cancers carrying ALK rearrangements. The recommended crizotinib dose was 250 mg twice daily in 28-day cycles.
In the overall NSCLC population a phase I trial investigated crizotinib in association to dose escalating erlotinib: 5 DLTs were reported (at 150/100 mg grade 2 vomiting, grade 2 esophagitis and dysphagia, grade 3 diarrhea and dehydration; at 200/100 mg, grade 3 dry eye and grade 3 esophagitis). Ninety-two percent of the patients experienced treatment-related AEs, mainly grade 1 or grade 2: diarrhea (72%), rash (56%) and fatigue (44%) (107).
Another phase I trials evaluated crizotinib in combination with dacomitinib, an irreversible pan-erb inhibitor in previously treated advanced NSCLC patients (108).
Cabozantinib
Cabozantinib (XL184) is a potent Met/VEGFR2/RET/KIT/AXL/FLT3 inhibitor that targets tumor survival, metastasization and angiogenesis.
It selectively inhibits KIT, RET, AXL, TIE2 and FLT3 (all kinases implicated in tumor pathobiology) through strong, reversible, ATP-competitive binding. Moreover, cabozantinib inhibits HGF and VEGF-mediated cell migration and also Met and VEGFR phosphorylation and the tubule formation, with no evidence of cytotoxicity.
This effect described in vitro, turned into in vivo significant tumor regression, without any relevant toxicity (109).
Several phase I trials have already been published verifying the schedule, the formulation, the dose of the drug, both as single-agent and in combination with other molecules.
Kurzrock and colleagues studied single-agent cabozantinib both in suspension and capsule formulation, at intermittent (5 days on, 9 off) and continuous schedule: MTD was defined at 175 mg continuous schedule, being DLT mucositis, elevated lipase and altered liver function (110).
The continuous dose was further investigated in a Japanese only population: MTD was 60 mg, being grade 3 hypertension the DLT (111).
Regarding combination regimens, a phase I study analyzed the interaction of the combination cabozantinib and rosiglitazone, as the latter is a CYP2C8 substrate, but no interaction was found between these two compounds (112,113).
Cabozantinib was further studied in several phase II trials in different tumor types. Among them, one phase II trial investigated treatment with cabozantinib in NSCLC patients previously treated with anti-EGFR TKi (50%) and anti-VEGF therapies (32%). At week 12 the ORR was 10% and the overall DCR 40%. No difference in terms of PFS (median 4.2 months) was seen in the two populations according to the treatment response at 12 weeks. The most common grade 3-4 events were diarrhea (7%), palmar-plantar erythrodyesthesia (8%), fatigue (13%) and asthenia (7%) (114).
Likewise tivantinib, also cabozantinib was tested together with erlotinib or gefitinib in vivo and in vitro in EGFR TKi resistant NSCLC xenograft models harboring Met amplification. Gefitinib and cabozantinib were tested on gefitinib resistant cell lines either alone and in combination and the two molecules together were substantially more potent than each drug alone (>50% inhibition). The same result was obtained with the combination of erlotinib and cabozantinib on erlotinib resistant cell lines (115).
The combination of cabozantinib and erlotinib was tested on 54 NSCLC patients in a phase Ib/II study. Patients were divided into 5 cohorts in two parallel arms: arm A (75 mg cabozantinib + 100 mg erlotinib; 125 mg cabozantinib + 100 mg erlotinib; 125 mg cabozantinib + 50 mg erlotinib) and arm B (75 mg cabozantinib +150 mg erlotinib; 50 mg cabozantinib +150 mg erlotinib). Twelve patients experienced at least 1 DLT: diarrhea, increased AST levels, palmar-plantar erythrodysesthesia, mucositis, hypertension, hypokalemia, elevated lipase and fatigue. The most common grade 3-4 adverse events were diarrhea (26%), fatigue (15%), dyspnea (12%) and hypoxia (9%) (116).
In advanced NSCLC patients two phase II trials are ongoing: the first one randomizes EGFR wild type patients to erlotinib, cabozantinib or erlotinib plus cabozantinib as second or third line therapy; the second study investigates cabozantinib in patients with KIF5B/RET positive NSCLC (117,118) (Table 1).
Foretinib
Foretinib (XL-880, EXEL-2880) is an oral multi-kinase inhibitor developed to target Met and several other receptor tyrosine kinases involved in tumor angiogenesis. It is an ATP-competitive inhibitor and binds the ATP pocket of both Met and VEGFR-2 tyrosine kinase domains with high affinity.
Both in vitro and in vivo, foretinib inhibits Met and VEGF receptor-2 (VEGFR-2) and have high in vitro affinity for PDGFRb, Tie-2, RON, Kit, and FLT3 kinases, preventing tumor growth through a direct effect on tumor cell proliferation and inhibition of invasion and angiogenesis, mediated by HGF and VEGF receptor (119).
Two phase I trials have been published: the first investigated foretinib administered for 5 consecutive days every 14 days in a 3+3 dose escalation study; in the second study foretinib was administered once daily for 28 days. Both trials were conducted in patients with metastatic or unresectable solid tumors. MDT was defined as 3.6 mg/kg for 5 consecutive days every 14 days in the first study and as 80 mg daily in the second; DLTs in the first study included grade 3 elevations in aspartate aminotransferase and lipase, whereas in the second trial hypertension, dehydration and diarrhea were described.
Additional AEs in both studies included hypertension, fatigue, diarrhea, vomiting, proteinuria, and hematuria. In these studies no responses were observed and most of patients achieved a stable disease as best response (120,121).
A phase I, open-label, randomized, 2-part crossover study assessed the safety, pharmacokinetics and relative bioavailability of single doses of foretinib free base tablet formulation compared to a bisphosphate salt capsule formulation: both were well tolerated and their pharmacokinetics and relative bioavailability were not clinically different (122).
On the basis of preclinical data, showing that combining foretinib with erlotinib or lapatinib effectively decrease the phosphorylation of Met, HER1, HER2, HER3, AKT, and ERK in cell lines, a phase I/II study of erlotinib in association or not with foretinib in previously treated NSCLC patients has been designed and is currently ongoing (123,124) (Table 1).
Golvatinib
Golvatinib (E7050) is a novel small molecule ATP-competitive inhibitor of Met receptor, that potently and selectively inhibits the autophosphorylation of Met and VEGF-induced phosphorylation of VEGFR (125).
Golvatinib also circumvents resistance to reversible, irreversible, and mutant-selective EGFR-TKIs induced by exogenous and/or endogenous HGF in EGFR mutant lung cancer cell lines, by blocking the Met/Gab1/PI3K/Akt pathway in vitro and also prevents the emergence of gefitinib-resistant cells, induced by continuous exposure to HGF (126).
A phase I study with oral daily golvatinib administered continuously once a day in patients with advanced solid tumors was performed. Three DLTs were observed: grade 3 increase in GGT and alkaline phosphatase levels and grade 3 fatigue, all at 450 mg. The MTD was determined to be 400 mg every day. Frequently occurring AEs were fatigue (68%), diarrhea (65%), nausea (62%), vomiting (53%), decreased appetite (47%), ALT increase (38%) and AST increase (23%). No grade 4 AEs were observed (127).
Other molecules
MGC D265 is an oral receptor tyrosine kinase inhibitor targeting Met, VEGF, RON and Tie2. Preclinical data have demonstrated synergism of action with erlotinib and early clinical trials are currently ongoing (128) (Table 1).
ANG707 is another non-selective Met inhibitor under investigation in early phase trials (129).
Antibodies
Antibodies against Met
Onartuzumab (MetMab)
MetMAb is a recombinant, fully humanized, monovalent monoclonal anti-Met antibody based on the human IgG1k framework sequence. It binds in the sema domain of Met within the extracellular domain, where it acts to inhibit HGF binding and initiation of receptor activation. The unique monovalent design of MetMAb eliminates the potential for Met activation via antibody-driven receptor dimerization (130).
The activity shown in vitro by MetMAb did not translate into a full activity in vivo: only about 65% tumor inhibition was demonstrated, indicating that blockade of HGF by MetMAb is not sufficient for full tumor inhibition in specific tumors (130).
A phase I trial investigated sequential 3+3 dose-escalation of endovenous MetMAb in advanced solid tumors: MetMAb was three weekly intravenously administered, both as single agent and in combination with bevacizumab 15 mg/kg every three weeks, until progression.
Most frequent MetMAb AEs as single-agent were: fatigue (56%), peripheral edema (35%), decreased appetite (32%), constipation (29%), nausea (27%), vomiting (24%) and hypoalbuminemia (24%); there was no consistent relationship between AEs and dose level.
Grade 3 AEs were peripheral edema (9%), abdominal pain, AST increase, fever and hyponatremia. No Grade 4 toxicity was observed. The combination arm had similar toxicities; no grade 3 or 4 toxicity was experienced. MTD was not reached. The best response was stable disease (131).
The phase II trial was a global, randomized, double-blind trial evaluating the combination of MetMAb + erlotinib versus placebo + erlotinib in second/third line NSCLC advanced patients. One hundred and twenty-eight NSCLC patients were enrolled with a baseline immunohistochemical evaluation of Met: 54% of the patients were considered as Met positive (high protein expression at IHC). Met positive patients treated in the experimental arm had a significantly higher PFS (3.0 vs. 1.5 months; HR 0.47; P=0.01) and OS (12.6 vs. 4.6 months; HR 0.37; P=0.002) (132).
Based on phase II data, a randomized, phase III, multicenter, double-blind, placebo-controlled study evaluating the efficacy and safety of onartuzumab in combination with erlotinib in patients with Met positive NSCLC who have received standard chemotherapy for advanced disease is currently recruiting patients (133) (Table 1).
The positive results of the phase I trial on MetMAb in combination with bevacizumab have paved the way to the ongoing randomized phase II multicentric double-blind placebo-controlled study evaluating the efficacy and safety of MetMAb in combination with either bevacizumab + platinum + paclitaxel or pemetrexed + platinum as first-line treatment in patients with stage IIIB and IV non-squamous NSCLC (134).
Antibodies against HGF
Ficlatuzumab
Ficlatuzumab (AV-299) is a potent hepatocyte growth factor (HGF) inhibitor IgG1 monoclonal antibody, that binds to the HGF ligand with high affinity and specificity. Ficlatuzumab was studied in two phase I trials and one phase II study. In both phase I trials it was associated with gefitinib and erlotinib. In the first phase I trial ficlatuzumab was biweekly administered intravenously over 30-60 minutes both as single-agent and in combination with erlotinib at 150 mg continuously in advanced solid tumors. There were no DLT in the monotherapy arm; consequently no MTD was identified.
For the combination arm there was one DLT (grade 3 mucositis). The RP2D for both monotherapy and combination regimen was defined as 20 mg/kg every 2 weeks. Ficlatuzumab as a single-agent demonstrated a stabilisation of disease in 50% of the cases (135).
The second phase Ib trial enrolled only Asiatic patients with unresectable NSCLC: ficlatuzumab was administered intravenously every 2 weeks at two dose levels (10 and 20 mg/kg) in combination with gefitinib at 250 mg daily. No DLTs were observed in the dose-escalation cohorts; 20 mg/kg of ficlatuzumab every 2 weeks + gefitinib 250 mg daily was selected as RP2D. Among 12 patients in the 20 mg/kg cohort, 5 partial responses were achieved (136). Most frequent treatment-emergent adverse events (AEs) were fatigue (27-33%), dermatitis acneiform (53%, particularly for the combination regimens), diarrhea (33-46%) and edema (16-27%) for both single-agent and combination therapy (135,136).
The efficacy of ficlatuzumab together with gefitinib was further investigated in a multicenter, open-label, exploratory, 2-arm randomized phase 2 study in previously untreated Asian NSCLC patients with the doses defined in the phase I. One-hundred eighty-eight patients were randomized with a baseline evaluation of Met by IHC and gene copy number. In the low Met group, ORR (41 versus 22%) and median PFS (7.3 versus 2.8 m) favored the combination regimen with a manageable toxicity profile (137).
Rilotumumab
Rilotumumab (AMG 102) is a fully human monoclonal antibody that selectively targets and neutralizes hepatocyte growth factor/scatter factor (HGF/SF). It preferentially bound to the β-chain of the human, mature, active form of HGF, and had no apparent effect on proteolytic processing of the inactive HGF precursor (138).
Two phase I trials have been published so far with AMG 102 in advanced refractory solid tumors: one as single agent and one in combination with bevacizumab or motesanib (139).
In the monotherapy trial, AMG 102 was well tolerated up to the planned maximum dose of 20 mg/kg, MTD was not reached and pharmacokinetic was linear. Two patients experienced DLTs: one grade 3 hypoxia and grade 3 dyspnea (0.5 mg/kg cohort) and one grade 3 upper GI hemorrhage (1 mg/kg cohort). Treatment-related AEs were generally mild and included fatigue (13%), constipation (8%), nausea (8%), vomiting (5%), anorexia (5%), myalgia (5%), and hypertension (5%). Seventy percent of the evaluable patients had a SD as best response (139).
The phase Ib combination study sequentially enrolled patients into four cohorts, but the number of those receiving AMG 102 plus motesanib was insufficient to adequately assess safety and the accrual was early suspended because of reports of cholecystitis in other motesanib studies. No dose-limiting toxicities were reported and the combination of AMG 102 with bevacizumab seemed to have acceptable toxicity. AEs were generally mild and included fatigue (75%), nausea (58%), constipation (42%) and peripheral edema (42%) (140).
TAK 701
TAK-701 is a humanized monoclonal antibody that binds HGF thus inhibiting its bound to Met receptor. TAK-701 in combination with gefitinib blocks the phosphorylation of Met, EGFR, extracellular signal-regulated kinase, and AKT in HGF expressing human NSCLC cell lines with an activating EGFR mutation. Combination therapy also markedly inhibited the tumor growth in vivo (141).
Preliminary data of a phase I study in advanced solid malignancies with TAK-701 showed that the most common AEs were cough, abdominal pain, constipation and fatigue, all grade 1-2. There were 3 grade 3 AEs (gastrointestinal ileus, pleural effusion, urinary tract infection) and 1 grade 4 AE (dyspnea). No DLT was found and the MTD has not been reached (142).
Conclusions
In patients with advanced NSCLC, a correct definition of the histotype is still the first step to design a proper therapeutic algorithm, but personalized molecular diagnosis is becoming more and more relevant.
Genetically defined subsets of cancers may share dependence on a specific signaling pathway: specific inhibitors targeting these pathways would be most effectively tested in patient populations characterized by molecular markers.
Moreover, genetic events that arise and are selected during tumor progression may become essential for tumor survival, a phenomenon generally described as “oncogene addiction”: cancer cells appear to depend on a single overactive oncogene to proliferate and survive (143). Optimal case selection, diagnostic and pharmacodynamic biomarker development, the identification and testing of rationally designed anticancer drugs and combination strategies are crucial to develop the best treatment for the right patient (144).
New generations of molecularly targeted drugs will allow more personalized medicine and more efficacious and less toxic antitumor therapies in patients with defined molecular aberrations, sparing normal cells thus sparing toxicity (145,146).
Met can act as an ‘oncogene expedient’ even in absence of genetic alterations and might potentiate the effect of other oncogenes, promote malignant progression and participate in tumor angiogenesis (147).
Met dysregulation correlates with disease prognosis in numerous cancers and represents a possible target for personalized treatment. The clinical efficacy of Met targeting agents in lung cancer needs further details from the ongoing trials as well as more information are necessary to establish the most appropriate diagnostic test to identify Met expression or amplification.
Several molecules are currently under investigation and two of them already reached phase III trials in advanced NSCLC.
Acknowledgements
Disclosure: All the authors have no conflict of interest to declare.
References
- Lynch TJ, Bell DW, Sordella R, et al. Activating mutations in the epidermal growth factor receptor underlying responsiveness of non-small-cell lung cancer to gefitinib. N Engl J Med 2004;350:2129-39.
- Mok TS, Wu YL, Thongprasert S, et al. Gefitinib or carboplatin-paclitaxel in pulmonary adenocarcinoma. N Engl J Med 2009;361:947-57.
- Mitsudomi T, Morita S, Yatabe Y, et al. Gefitinib versus cisplatin plus docetaxel in patients with non-small-cell lung cancer harbouring mutations of the epidermal growth factor receptor (WJTOG3405): an open label, randomised phase 3 trial. Lancet Oncol 2010;11:121-8.
- Maemondo M, Inoue A, Kobayashi K, et al. Gefitinib or chemotherapy for non-small-cell lung cancer with mutated EGFR. N Engl J Med 2010;362:2380-8.
- Zhou C, Wu YL, Chen G, et al. Erlotinib versus chemotherapy as first-line treatment for patients with advanced EGFR mutation-positive non-small-cell lung cancer (OPTIMAL, CTONG-0802): a multicentre, open-label, randomised, phase 3 study. Lancet Oncol 2011;12:735-42.
- Rosell R, Carcereny E, Gervais R, et al. Erlotinib versus standard chemotherapy as first-line treatment for European patients with advanced EGFR mutation-positive non-small-cell lung cancer (EURTAC): a multicentre, open-label, randomised phase 3 trial. Lancet Oncol 2012;13:239-46.
- Yang JC, Schuler MH, Yamamoto N, et al. LUX-Lung 3: A randomized, open-label, phase III study of afatinib versus pemetrexed and cisplatin as first-line treatment for patients with advanced adenocarcinoma of the lung harboring EGFR-activating mutations. J Clin Oncol 2012;30:abstr LBA7500.
- Morris SW, Kirstein MN, Valentine MB, et al. Fusion of a kinase gene, ALK, to a nucleolar protein gene, NPM, in non-Hodgkin’s lymphoma. Science 1994;263:1281-4.
- Soda M, Choi YL, Enomoto M, et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 2007;448:561-6.
- Kwak EL, Bang YJ, Camidge DR, et al. Anaplastic lymphoma kinase inhibition in non-small-cell lung cancer. N Engl J Med 2010;363:1693-703.
- Shaw AT, Kim DW, Nakagawa K, et al. Phase III study of crizotinib versus pemetrexed or docetaxel chemotherapy in patients with advanced ALK-positive non-small cell lung cancer (NSCLC) (PROFILE 1007). Ann Oncology 2012;23:abstr LBA1_PR.
- Lord RV, Brabender J, Gandara D, et al. Low ERCC1 expression correlates with prolonged survival after cisplatin plus gemcitabine chemotherapy in non-small cell lung cancer. Clin Cancer Res 2002;8:2286-91.
- Tsao MS, Aviel-Ronen S, Ding K, et al. Prognostic and predictive importance of p53 and RAS for adjuvant chemotherapy in non small-cell lung cancer. J Clin Oncol 2007;25:5240-7.
- Huncharek M, Muscat J, Geschwind JF. K-ras oncogene mutation as a prognostic marker in non-small cell lung cancer: a combined analysis of 881 cases. Carcinogenesis 1999;20:1507-10.
- Eberhard DA, Johnson BE, Amler LC, et al. Mutations in the epidermal growth factor receptor and in KRAS are predictive and prognostic indicators in patients with non-small-cell lung cancer treated with chemotherapy alone and in combination with erlotinib. J Clin Oncol 2005;23:5900-9.
- Janne PA, Shaw AT, Pereira JR, et al. Phase II doubleblind, randomized study of selumetinib (SEL) plus docetaxel (DOC) versus DOC plus placebo as second-line treatment for advanced KRAS mutant non-small cell lung cancer (NSCLC). J Clin Oncol 2012;30:abstr 7503.
- Jimenez C, Jones DR, Rodríguez-Viciana P, et al. Identification and characterization of a new oncogene derived from the regulatory subunit of phosphoinositide 3-kinase. EMBO J 1998;17:743-53.
- Samuels Y, Wang Z, Bardelli A, et al. High frequency of mutations of the PIK3CA gene in human cancers. Science 2004;304:554.
- Stephens P, Hunter C, Bignell G, et al. Lung cancer: intragenic ERBB2 kinase mutations in tumours. Nature 2004;431:525-6.
- Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature 2002;417:949-54.
- Chin LP, Soo RA, Soong R, et al. Targeting ROS1 with anaplastic lymphoma kinase inhibitors: a promising therapeutic strategy for a newly defined molecular subset of non-small-cell lung cancer. J Thorac Oncol 2012;7:1625-30.
- Cooper CS, Park M, Blair DG, et al. Molecular cloning of a new transforming gene from a chemically transformed human cell line. Nature 1984;311:29-33.
- Park M, Dean M, Kaul K, et al. Sequence of MET protooncogene cDNA has features characteristic of the tyrosine kinase family of growth-factor receptors. Proc Natl Acad Sci U S A 1987;84:6379-83.
- Bottaro DP, Rubin JS, Faletto DL, et al. Identification of the hepatocyte growth factor receptor as the c-met protooncogene product. Science 1991;251:802-4.
- Birchmeier C, Gherardi E. Developmental roles of HGF/ SF and its receptor, the c-Met tyrosine kinase. Trends Cell Biol 1998;8:404-10.
- Comoglio PM. Pathway specificity for Met signalling. Nat Cell Biol 2001;3:E161-2.
- Ma PC, Kijima T, Maulik G, et al. c-MET mutational analysis in small cell lung cancer: novel juxtamembrane domain mutations regulating cytoskeletal functions. Cancer Res 2003;63:6272-81.
- Jeffers M, Rong S, Vande Woude GF. Enhanced tumorigenicity and invasion-metastasis by hepatocyte growth factor/scatter factor-met signalling in human cells concomitant with induction of the urokinase proteolysis network. Mol Cell Biol 1996;16:1115-25.
- Cecchi F, Rabe DC, Bottaro DP. Targeting the HGF/Met signalling pathway in cancer. Eur J Cancer 2010;46:1260-70.
- Ponzetto C, Bardelli A, Zhen Z, et al. A multifunctional docking site mediates signaling and transformation by the hepatocyte growth factor/scatter factor receptor family. Cell 1994;77:261-71.
- Birchmeier C, Birchmeier W, Gherardi E, et al. Met, metastasis, motility and more. Nat Rev Mol Cell Biol 2003;4:915-25.
- Gohda E, Tsubouchi H, Nakayama H, et al. Purification and partial characterization of hepatocyte growth factor from plasma of a patient with fulminant hepatic failure. J Clin Invest 1988;81:414-9.
- Montesano R, Matsumoto K, Nakamura T, et al. Identification of a fibroblast-derived epithelial morphogen as hepatocyte growth factor. Cell 1991;67:901-8.
- Rosen EM, Nigam SK, Goldberg ID. Scatter factor and the c-met receptor: a paradigm for mesenchymal/epithelial interaction. J Cell Biol 1994;127:1783-7.
- Hammond DE, Urbé S, Vande Woude GF, et al. Downregulation of MET, the receptor for hepatocyte growth factor. Oncogene 2001;20:2761-70.
- Goldoni S, Humphries A, Nyström A, et al. Decorin is a novel antagonistic ligand of the Met receptor. J Cell Biol 2009;185:743-54.
- Rong S, Segal S, Anver M, et al. Invasiveness and metastasis of NIH 3T3 cells induced by Met-hepatocyte growth factor/scatter factor autocrine stimulation. Proc Natl Acad Sci U S A 1994;91:4731-5.
- Boccaccio C, Sabatino G, Medico E, et al. The MET oncogene drives a genetic programme linking cancer to haemostasis. Nature 2005;434:396-400.
- Takayama H, LaRochelle WJ, Sharp R, et al. Diverse tumorigenesis associated with aberrant development in mice overexpressing hepatocyte growth factor/scatter factor. Proc Natl Acad Sci U S A 1997;94:701-6.
- Abounader R, Lal B, Luddy C, et al. In vivo targeting of SF/HGF and c-met expression via U1snRNA/ribozymes inhibits glioma growth and angiogenesis and promotes apoptosis. FASEB J 2002;16:108-10.
- Eder JP, Vande Woude GF, Boerner SA, et al. Novel therapeutic inhibitors of the c-Met signaling pathway in cancer. Clin Cancer Res 2009;15:2207-14.
- Shojaei F, Lee JH, Simmons BH, et al. HGF/c-Met acts as an alternative angiogenic pathway in sunitinib-resistant tumors. Cancer Res 2010;70:10090-100.
- Pennacchietti S, Michieli P, Galluzzo M, et al. Hypoxia promotes invasive growth by transcriptional activation of the met protooncogene. Cancer Cell 2003;3:347-61.
- De Bacco F, Luraghi P, Medico E, et al. Induction of MET by ionizing radiation and its role in radioresistance and invasive growth of cancer. J Natl Cancer Inst 2011;103:645-61.
- Lai AZ, Abella JV, Park M. Crosstalk in Met receptor oncogenesis. Trends Cell Biol 2009;19:542-51.
- Jo M, Stolz DB, Esplen JE, et al. Cross-talk between epidermal growth factor receptor and c-Met signal pathways in transformed cells. J Biol Chem 2000;275:8806-11.
- Stommel JM, Kimmelman AC, Ying H, et al. Coactivation of receptor tyrosine kinases affects the response of tumor cells to targeted therapies. Science 2007;318:287-90.
- Guo A, Villén J, Kornhauser J, et al. Signaling networks assembled by oncogenic EGFR and c-Met. Proc Natl Acad Sci U S A 2008;105:692-7.
- Tang Z, Du R, Jiang S, et al. Dual MET-EGFR combinatorial inhibition against T790M-EGFRmediated erlotinib-resistant lung cancer. Br J Cancer 2008;99:911-22.
- Shattuck DL, Miller JK, Carraway KL 3rd, et al. Met receptor contributes to trastuzumab resistance of Her2-overexpressing breast cancer cells. Cancer Res 2008;68:1471-7.
- Nakajima M, Sawada H, Yamada Y, et al. The prognostic significance of amplification and overexpression of c-met and c-erb B-2 in human gastric carcinomas. Cancer 1999;85:1894-902.
- Mueller KL, Hunter LA, Ethier SP, et al. Met and c-Src cooperate to compensate for loss of epidermal growth factor receptor kinase activity in breast cancer cells. Cancer Res 2008;68:3314-22.
- Engelman JA, Zejnullahu K, Mitsudomi T, et al. MET amplification leads to gefitinib resistance in lung cancer by activating ERBB3 signaling. Science 2007;316:1039-43.
- Cappuzzo F, Jänne PA, Skokan M, et al. MET increased gene copy number and primary resistance to gefitinib therapy in non-small-cell lung cancer patients. Ann Oncol 2009;20:298-304.
- Swiercz JM, Worzfeld T, Offermanns S. ErbB-2 and met reciprocally regulate cellular signaling via plexin-B1. J Biol Chem 2008;283:1893-901.
- Khoury H, Naujokas MA, Zuo D, et al. HGF converts ErbB2/Neu epithelial morphogenesis to cell invasion. Mol Biol Cell 2005;16:550-61.
- Matsubara D, Ishikawa S, Oguni S, et al. Molecular predictors of sensitivity to the MET inhibitor PHA665752 in lung carcinoma cells. J Thorac Oncol 2010;5:1317-24.
- Turke AB, Zejnullahu K, Wu YL, et al. Preexistence and clonal selection of MET amplification in EGFR mutant NSCLC. Cancer Cell 2010;17:77-88.
- Goldoni S, Iozzo RV. Tumor microenvironment: Modulation by decorin and related molecules harboring leucine-rich tandem motifs. Int J Cancer 2008;123:2473-9.
- Gur G, Rubin C, Katz M, et al. LRIG1 restricts growth factor signaling by enhancing receptor ubiquitylation and degradation. EMBO J 2004;23:3270-81.
- Long IS, Han K, Li M, et al. Met receptor overexpression and oncogenic Ki-ras mutation cooperate to enhance tumorigenicity of colon cancer cells in vivo. Mol Cancer Res 2003;1:393-401.
- Ma PC, Jagadeeswaran R, Jagadeesh S, et al. Functional expression and mutations of c-Met and its therapeutic inhibition with SU11274 and small interfering RNA in non-small cell lung cancer. Cancer Res 2005;65:1479-88.
- Pelicci G, Giordano S, Zhen Z, et al. The motogenic and mitogenic responses to HGF are amplified by the Shc adaptor protein. Oncogene 1995;10:1631-8.
- Wang X, DeFrances MC, Dai Y, et al. A mechanism of cell survival: sequestration of Fas by the HGF receptor Met. Mol Cell 2002;9:411-21.
- Olivero M, Rizzo M, Madeddu R, et al. Overexpression and activation of hepatocyte growth factor/scatter factor in human non-small-cell lung carcinomas. Br J Cancer 1996;74:1862-8.
- Maulik G, Kijima T, Ma PC, et al. Modulation of the c-Met/hepatocyte growth factor pathway in small cell lung cancer. Clin Cancer Res 2002;8:620-7.
- Ma PC, Tretiakova MS, MacKinnon AC, et al. Expression and mutational analysis of MET in human solid cancers. Genes Chromosomes Cancer 2008;47:1025-37.
- Lutterbach B, Zeng Q, Davis LJ, et al. Lung cancer cell lines harboring MET gene amplification are dependent on Met for growth and survival. Cancer Res 2007;67:2081-8.
- Beau-Faller M, Ruppert AM, Voegeli AC, et al. MET gene copy number in non-small cell lung cancer: molecular analysis in a targeted tyrosine kinase inhibitor naïve cohort. J Thorac Oncol 2008;3:331-9.
- Kanteti R, Yala S, Ferguson MK, et al. MET, HGF, EGFR, and PXN gene copy number in lung cancer using DNA extracts from FFPE archival samples and prognostic significance. J Environ Pathol Toxicol Oncol 2009;28:89-98.
- Onozato R, Kosaka T, Kuwano H, et al. Activation of MET by gene amplification or by splice mutations deleting the juxtamembrane domain in primary resected lung cancers. J Thorac Oncol 2009;4:5-11.
- Onitsuka T, Uramoto H, Ono K, et al. Comprehensive molecular analyses of lung adenocarcinoma with regard to the epidermal growth factor receptor, K-ras, MET, and hepatocyte growth factor status. J Thorac Oncol 2010;5:591-6.
- Kong-Beltran M, Seshagiri S, Zha J, et al. Somatic mutations lead to an oncogenic deletion of met in lung cancer. Cancer Res 2006;66:283-9.
- Di Renzo MF, Olivero M, Martone T, et al. Somatic mutations of the MET oncogene are selected during metastatic spread of human HNSC carcinomas. Oncogene 2000;19:1547-55.
- Cheng TL, Chang MY, Huang SY, et al. Overexpression of circulating c-met messenger RNA is significantly correlated with nodal stage and early recurrence in nonsmall cell lung cancer. Chest 2005;128:1453-60.
- Go H, Jeon YK, Park HJ, et al. High MET gene copy number leads to shorter survival in patients with non-small cell lung cancer. J Thorac Oncol 2010;5:305-13.
- Okuda K, Sasaki H, Yukiue H, et al. Met gene copy number predicts the prognosis for completely resected non-small cell lung cancer. Cancer Sci 2008;99:2280-5.
- Cappuzzo F, Jänne PA, Skokan M, et al. MET increased gene copy number and primary resistance to gefitinib therapy in non-small-cell lung cancer patients. Ann Oncol 2009;20:298-304.
- Bean J, Brennan C, Shih JY, et al. MET amplification occurs with or without T790M mutations in EGFR mutant lung tumors with acquired resistance to gefitinib or erlotinib. Proc Natl Acad Sci U S A 2007;104:20932-7.
- Cappuzzo F, Marchetti A, Skokan M, et al. Increased MET gene copy number negatively affects survival of surgically resected non-small-cell lung cancer patients. J Clin Oncol 2009;27:1667-74.
- Rho JK, Choi YJ, Jeon BS, et al. Combined treatment with silibinin and epidermal growth factor receptor tyrosine kinase inhibitors overcomes drug resistance caused by T790M mutation. Mol Cancer Ther 2010;9:3233-43.
- Arcila ME, Oxnard GR, Nafa K, et al. Rebiopsy of lung cancer patients with acquired resistance to EGFR inhibitors and enhanced detection of the T790M mutation using a locked nucleic acid-based assay. Clin Cancer Res 2011;17:1169-80.
- Sequist LV, Waltman BA, Dias-Santagata D, et al. Genotypic and histological evolution of lung cancers acquiring resistance to EGFR inhibitors. Sci Transl Med 2011;3:75ra26.
- Tomoko K, Kadofuku T, Yamaoka T, et al. The novel c-MET inhibitor, ARQ 197, shows additive growthinhibitory effect with erlotinib through enhanced degradation of c-MET protein via ubiquitin/proteasome pathway. Cancer Res 2010;70:abstr1639.
- Munshi N, Jeay S, Li Y, et al. ARQ 197, a novel and selective inhibitor of the human c-Met receptor tyrosine kinase with antitumor activity. Mol Cancer Ther 2010;9:1544-53.
- Chen CR, Szwaya J, Rajanackarin A, et al. Combination studies of tyrosine kinase inhibitors (TKIs): assessment of potential cytotoxic synergy of ARQ 197 with sorafenib or sunitinib. AACR Meeting 2009; abstr 820.
- Jeay S, Munshi N, Hill J, et al. ARQ 197, a highly selective small molecule inhibitor of c-Met, with selective antitumor properties in a broad spectrum of human cancer cells. AACR Annual Meeting 2007; abstr 2369.
- Yap TA, Olmos D, Brunetto AT, et al. Phase I trial of a selective c-MET inhibitor ARQ 197 incorporating proof of mechanism pharmacodynamic studies. J Clin Oncol 2011;29:1271-9.
- Rosen LS, Senzer N, Mekhail T, et al. A phase I doseescalation study of Tivantinib (ARQ 197) in adult patients with metastatic solid tumors. Clin Cancer Res 2011;17:7754-64.
- Rodriguez-Antona C, Ingelman-Sundberg M. Cytochrome P450 pharmacogenetics and cancer. Oncogene 2006;25:1679-91.
- Nishina T, Hirashima T, Sugio K, et al. The effect of CYP2C19 polymorphism on the tolerability of ARQ 197: Results from phase I trial in Japanese patients with metastatic solid tumors. J Clin Oncol 2011;29:abstr 2516.
- NCT01377376; Available online: www.clinicaltrial.gov
- Available online: http://propthink.com/arqule-setbacksincrease- risk-discount-rates-increased-for-forwardrevenue/ 1766
- Goldman JW, Laux I, Chai F, et al. Phase 1 doseescalation trial evaluating the combination of the selective MET (mesenchymal-epithelial transition factor) inhibitor tivantinib (ARQ 197) plus erlotinib. Cancer 2012;118:5903-11.
- Sequist LV, von Pawel J, Garmey EG, et al. Randomized phase II study of erlotinib plus tivantinib versus erlotinib plus placebo in previously treated non-small-cell lung cancer. J Clin Oncol 2011;29:3307-15.
- Scagliotti GV, Novello S, Schiller JH, et al. Rationale and design of MARQUEE: a phase III, randomized, doubleblind study of tivantinib plus erlotinib versus placebo plus erlotinib in previously treated patients with locally advanced or metastatic, nonsquamous, non-small-cell lung cancer. Clin Lung Cancer 2012;13:391-5.
- Available online: http://propthink.com/tivantinib-will-notmeet- primary-endpoint-phase-iii-trial-discontinued/2390
- Zou HY, Li Q, Lee JH, et al. Sensitivity of selected human tumor models to PF-04217903, a novel selective c-Met kinase inhibitor. Mol Cancer Ther 2012;11:1036-47.
- NCT00706355. Available online: www.clinicaltrial.gov
- NCT01253707. Available online: www.clinicaltrial.gov
- Koblish HK, Liu X, Hall L, et al. Preclinical in vivo characterization of INCB028060, a novel, potent and highly selective c-Met inhibitor. J Clin Oncol 2008;26:abstr 14561.
- Liu X, Wang Q, Yang G, et al. A novel kinase inhibitor, INCB28060, blocks c-MET-dependent signaling, neoplastic activities, and cross-talk with EGFR and HER- 3. Clin Cancer Res 2011;17:7127-38.
- Donehower RC, Scardina A, Hill M, et al. A phase I doseescalation study of INCB028060, an inhibitor of c-MET receptor tyrosine kinase, in patients with advanced solid tumors. J Clin Oncol 2011;29:abstr 3091.
- Cui JJ, Tran-Dubé M, Shen H, et al. Structure based drug design of crizotinib (PF-02341066), a potent and selective dual inhibitor of mesenchymal-epithelial transition factor (c-MET) kinase and anaplastic lymphoma kinase (ALK). J Med Chem 2011;54:6342-63.
- Tanizaki J, Okamoto I, Okamoto K, et al. MET tyrosine kinase inhibitor crizotinib (PF-02341066) shows differential antitumor effects in non-small cell lung cancer according to MET alterations. J Thorac Oncol 2011;6:1624-31.
- Ou SH, Kwak EL, Siwak-Tapp C, et al. Activity of crizotinib (PF02341066), a dual mesenchymal-epithelial transition (MET) and anaplastic lymphoma kinase (ALK) inhibitor, in a non-small cell lung cancer patient with de novo MET amplification. J Thorac Oncol 2011;6:942-6.
- Ou SH, Govindan R, Eaton KD, et al. Phase I/II dosefinding study of crizotinib (CRIZ) in combination with erlotinib (E) in patients (pts) with advanced non-small cell lung cancer (NSCLC). J Clin Oncol 2012;30:abstr 2610.
- Janne PA, Shaw AT, Giaccone G, et al. Phase I trial of irreversible pan-erbb inhibitor dacomitinib (dac) in combination with ALK/MET inhibitor crizotinib (criz) in previously treated advanced non-small cell lung cancer (NSCLC). Annals of Oncology 2012;23:abstr 1290P.
- Yakes FM, Chen J, Tan J, et al. Cabozantinib (XL184), a novel MET and VEGFR2 inhibitor, simultaneously suppresses metastasis, angiogenesis, and tumor growth. Mol Cancer Ther 2011;10:2298-308.
- Kurzrock R, Sherman SI, Ball DW, et al. Activity of XL184 (Cabozantinib), an oral tyrosine kinase inhibitor, in patients with medullary thyroid cancer. J Clin Oncol 2011;29:2660-6.
- Nokihara H, Yamamoto N, Nakamichi S, et al. Molecular profile and anti-tumor activity in nonsmallcell lung cancer (NSCLC) patients (pts) in a phase 1 study of cabozantinib (XL184) in Japan. ESMO Meeting 2012; Ann Oncol 2012;23:abstr 17O8PD.
- Choueiri TK, Pal SK, Cabanillas ME, et al. Antitumor activity observed in a phase I drug-drug interaction study of cabozantinib (XL184) and rosiglitazone in patients (pts) with renal cell carcinoma (RCC) and differentiated thyroid cancer (DTC). J Clin Oncol 2011;29:abstr e13042.
- Choueiri TK, Pal SK, McDermott DF, et al. Efficacy of cabozantinib (XL184) in patients (pts) with metastatic, refractory renal cell carcinoma (RCC). J Clin Oncol 2012;30:abstr 4504.
- Hellerstedt BA, Edelman G, Vogelzang NJ, et al. Activity of cabozantinib (XL184) in metastatic NSCLC: Results from a phase II randomized discontinuation trial (RDT). J Clin Oncol 2012;30:abstr 7514.
- Jänne PA, Wax M, Leach J, et al. Targeting MET with XL 184 to reverse EGFR tyrosine kinase inhibitor (TKI) resistance in NSCLC: impact of preclinical studies on clinical trial design. 20th EORTC-NCIAACR Symposium on Molecular Targets and Cancer Therapeutics, poster 552.
- Wakelee HA, Gettinger SN, Engelman JA, et al. A phase Ib/II study of XL184 (BMS 907351) with and without erlotinib in patients (pts) with non-small cell lung cancer (NSCLC). J Clin Oncol 2010;28:abstr 3017.
- NCT01708954. Available online: www.clinicaltrial.gov
- NCT01639508. Available online: www.clinicaltrial.gov
- Qian F, Engst S, Yamaguchi K, et al. Inhibition of tumor cell growth, invasion, and metastasis by EXEL- 2880 (XL880, GSK1363089), a novel inhibitor of HGF and VEGF receptor tyrosine kinases. Cancer Res 2009;69:8009-16.
- Eder JP, Shapiro GI, Appleman LJ, et al. A phase I study of foretinib, a multi-targeted inhibitor of c-Met and vascular endothelial growth factor receptor 2. Clin Cancer Res 2010;16:3507-16.
- Shapiro GI, McCallum S, Adams LM, et al. A Phase 1 dose-escalation study of the safety and pharmacokinetics of once-daily oral foretinib, a multi-kinase inhibitor, in patients with solid tumors. Invest New Drugs 2012. [Epub ahead of print].
- Naing A, Kurzrock R, Adams LM, et al. A comparison of the pharmacokinetics of the anticancer MET inhibitor foretinib free base tablet formulation to bisphosphate salt capsule formulation in patients with solid tumors. Invest New Drugs 2012;30:327-34.
- Liu L, Shi H, Liu Y, Anderson A, et al. Synergistic effects of foretinib with HER-targeted agents in MET and HER1- or HER2-coactivated tumor cells. Mol Cancer Ther 2011;10:518-30.
- NCT01068587. Available online: www.clinicaltrial.gov
- Nakagawa T, Tohyama O, Yamaguchi A, et al. E7050: a dual c-Met and VEGFR-2 tyrosine kinase inhibitor promotes tumor regression and prolongs survival in mouse xenograft models. Cancer Sci 2010;101:210-5.
- Wang W, Li Q, Takeuchi S, et al. Met kinase inhibitor E7050 reverses three different mechanisms of hepatocyte growth factor-induced tyrosine kinase inhibitor resistance in EGFR mutant lung cancer. Clin Cancer Res 2012;18:1663-71.
- Daniele G, Ranson M, Blanco-Codesido M, et al. Phase I dose-finding study of golvatinib (E7050), a c-Met and Eph receptor targeted multi-kinase inhibitor, administered orally QD to patients with advanced solid tumors. J Clin Oncol 2012;30:abstr 3030.
- Bonfils C, Beaulieu N, Fournel M, et al. The combination of MGC D265, a MET/VEGFR inhibitor in clinical development, and erlotinib potently inhibits tumour growth by altering multiple pathways including glycolysis. AACR 2012, abstr 1790.
- Peters S, Adjei AA. MET: a promising anticancer therapeutic target. Nat Rev Clin Oncol 2012;9:314-26.
- Prat M, Crepaldi T, Pennacchietti S, et al. Agonistic monoclonal antibodies against the Met receptor dissect the biological responses to HGF. J Cell Sci 1998;111:237-47.
- Moss RA, Bothos J, Patel P, et al. Complete results from phase I dose escalation study of MetMAb, a monovalent antagonist antibody to the receptor met, dosed as single agent and in combination with bevacizumab in patients with advanced solid malignancies. Ann Oncol 2010;21:abstr 504P.
- Spigel DR, Ervin TJ, Ramlau R, et al. Final efficacy results from OAM4558g, a randomized phase II study evaluating MetMAb or placebo in combination with erlotinib in advanced NSCLC. J Clin Oncol 2011;29:abstr 7505.
- NCT01496742. Available online: www.clinicaltrial.gov
- NCT01456325. Available online: www.clinicaltrial.gov
- Patnaik A, Weiss GJ, Papadopoulos K, et al. Phase I study of SCH 900105 (SC), an anti-hepatocyte growth factor (HGF) monoclonal antibody (MAb), as a single agent and in combination with erlotinib (E) in patients (pts) with advanced solid tumors. J Clin Oncol 2010; 28:abstr 2525.
- Tan E, Park K, Lim W, et al. A Phase 1b Study of Ficlatuzumab (AV-299), an anti-hepatocyte growth factor monoclonal antibody, in combination with gefitinib in asian patients with NSCLC. J Clin Oncol 2011;29:abstr 7571.
- Mok TS, Park K, Geater SL, et al. A randomized phase 2 study with exploratory biomarker analysis of ficlatuzumab a humanized hepatocyte growth factor inhibitory MAB in combination with gefitinib versus gefitinib in Asian patients with lung adenocarcinoma. Ann Oncol 2012;23:abstr 1198P.
- Burgess TL, Sun J, Meyer S, et al. Biochemical characterization of AMG 102: a neutralizing, fully human monoclonal antibody to human and nonhuman primate hepatocyte growth factor. Mol Cancer Ther 2010;9:400-9.
- Gordon MS, Sweeney CS, Mendelson DS, et al. Safety, pharmacokinetics, and pharmacodynamics of AMG 102, a fully human hepatocyte growth factor-neutralizing monoclonal antibody, in a first-in-human study of patients with advanced solid tumors. Clin Cancer Res 2010;16:699-710.
- Rosen PJ, Sweeney CJ, Park DJ, et al. A phase Ib study of AMG 102 in combination with bevacizumab or motesanib in patients with advanced solid tumors. Clin Cancer Res 2010;16:2677-87.
- Okamoto W, Okamoto I, Tanaka K, et al. TAK-701, a humanized monoclonal antibody to hepatocyte growth factor, reverses gefitinib resistance induced by tumorderived HGF in non-small cell lung cancer with an EGFR mutation. Mol Cancer Ther 2010;9:2785-92.
- Jones SF, Cohen RB, Bendell JC, et al. Safety, tolerability, and pharmacokinetics of TAK-701, a humanized antihepatocyte growth factor (HGF) monoclonal antibody, in patients with advanced non hematologic malignancies: First-in-human phase I dose-escalation study. J Clin Oncol 2010; 28:abstr 3081.
- Sharma SV, Settleman J. Oncogene addiction: setting the stage for molecularly targeted cancer therapy. Genes Dev 2007;21:3214-31.
- de Bono JS, Ashworth A. Translating cancer research into targeted therapeutics. Nature 2010;467:543-9.
- de Bono JS, Tolcher AW, Rowinsky EK. The future of cytotoxic therapy: selective cytotoxicity based on biology is the key. Breast Cancer Res 2003;5:154-9.
- de Bono JS, Yap TA. Future directions in the evaluation of c-MET-driven malignancies. Ther Adv Med Oncol 2011;3:S51-60.
- Comoglio PM, Giordano S, Trusolino L. Drug development of MET inhibitors: targeting oncogene addiction and expedience. Nat Rev Drug Discov 2008;7:504-16.