
Empiric Radiotherapy for Lung Cancer Collaborative Group multi-institutional evidence-based guidelines for the use of empiric stereotactic body radiation therapy for non-small cell lung cancer without pathologic confirmation
Introduction
The standard treatment of early-stage non-small cell lung cancer (NSCLC) has been surgical resection when feasible. However, in patients who are surgically or medically inoperable or by patient preference, stereotactic body radiation therapy (SBRT) has become established as an effective definitive modality to treat stage I NSCLC (1), and it is emerging as a viable modality for medically operable patients (2). Many patients treated with SBRT have poor pulmonary reserve and significant co-morbidities and are, therefore, also at significant risk of developing complications from biopsy. While a historical resolute mantra of cancer care is that no therapy is administered without pathologic proof of malignancy, clinicians are increasingly faced with the challenge of considering SBRT to be performed empirically for patients with radiographic findings consistent with early-stage lung cancer. We performed a thorough literature search and formed the Empiric Radiotherapy for Lung Cancer Collaborative Group to report the evidence regarding empiric SBRT and guidelines for physicians to consider. Herein, we provide an expert commentary on how to apply “rationalism to empiricism,” or when it is appropriate to treat a patient with SBRT without pathologic confirmation.
Scope of the problem
Lung cancer is the leading cause of cancer-related death in the United States in both men and women, with an estimated 154,050 deaths annually (3). The National Lung Screening Trial (NLST) randomized asymptomatic patients at high risk for lung cancer to either low-dose CT (LDCT) or chest radiography and found that LDCT identified more cases of lung cancer (1.1% vs. 0.7%) with a sensitivity and specificity of 93.8% and 73.4%, respectively (4). This study led to numerous professional organizations rapidly endorsing screening, including the United States Preventative Services Task Force (USPSTF) (5). Despite this recommendation, lung cancer screening is only slowly being adopted due to concerns over resources, radiation dose, and cost-effectiveness (5). The question of when to perform empiric SBRT will become increasingly critical in the era of screening (6-9). Also important is to recognize that false positive results were noted in 96.4% of the positive screening results in the LDCT group of the NLST. As such, there is great need to establish guidelines to improve the predictive yield of an abnormal screening result, with special reference to patients at high risk of invasive diagnostic complications and hence referred for empiric SBRT.
Indications for SBRT and accompanying risks and benefits
SBRT has emerged as a standard of care for inoperable stage I NSCLC with highly effective control of the primary tumor, but, like surgery, it similarly carries a challenging 5-year disease free survival of 45–63% (10-13). A pooled analysis of two randomized trials (STARS, ROSEL) of SBRT versus surgery in operable patients found equal relapse-free survival, although overall survival (OS) was improved in the SBRT arm potentially due to excessive toxicities associated with surgery (14). Therefore, while the most accepted use of SBRT is in the inoperable population, SBRT is increasingly being considered in the operable population as additional trials in medically operable patients are reported (15).
SBRT, while generally very well tolerated, carries a low risk of adverse events (16), including pneumonitis (17), chest wall pain syndrome and rib fracture for peripheral lesions (18), and concern for great vessel or airway injury for central tumors, defined as within 2 cm of the tracheobronchial tree (19). The concern for potentially lethal toxicity is greatest for central lesions and especially ultra-central lesions (20), although recent evidence supports safe fractionation for central lesions (21). Therefore, location can be one consideration for whether or not to perform empiric SBRT. It is also important to note that many patients with central or ultra-central lesions who are at a higher risk of toxicities from radiation are also not candidates for sub-lobar resections. SBRT, therefore, is an important aspect of curative intent treatment for these patients, with or without pathological diagnosis. In these guidelines, we only discuss the management of non-central lesions, as the risk-benefit ratio will be defined with evolving results from the prospective NRG Oncology RTOG 0813 trial.
With increasing diagnoses of early stage lung cancer, clinicians are often faced with the question of surgery without a biopsy or empiric SBRT. Surgery provides a pathologic diagnosis (PD) and simultaneous lymph node analysis, and this information can guide therapeutic decision making such as conducting molecular analysis and need for adjuvant therapy. Most importantly, the vast majority of lung nodules identified are not lung cancer, with the positive predictive value in a high-risk population of the NLST of only 2.4% (4). Therefore, overtreatment of benign nodules with SBRT remains a potential concern. Another common clinical scenario is for patients who have undergone a prior surgical (lobar or sub-lobar) resection or SBRT with preceding tissue confirmation and are noted to have serial enlargement or development of a new lung nodule radiographically consistent with a new primary lung cancer. In this setting, proceeding with SBRT without an attempted biopsy in select patients can be a reasonable option since the risks of biopsy may be higher in patients having undergone prior surgery and the likelihood of cancer may be higher given the prior personal history of lung cancer in these patients.
In patients not undergoing definitive resection, histologic confirmation can be obtained via a transthoracic computed tomography-guided needle biopsy or via a transbronchial biopsy, which allows for simultaneous nodal evaluation via endobronchial ultrasound (EBUS) (Table 1). In addition to providing definitive diagnostic confirmation and staging, tissue diagnosis also may also allow for prognostication between NSCLC histologies (22) and between NSCLC and early stage small cell lung cancer (23) which can guide subsequent decisions for systemic therapy (24). However, arguments can be made against obtaining a pre-treatment histologic diagnosis. First, transthoracic computed tomography-guided needle biopsy carries approximately a 20% risk of pneumothorax and a 5% risk of serious complications such as chest tube placement. These risks can be as high as 35% and 10.7%, respectively, in a typical medically inoperable patient or those with limited pulmonary reserves (25). The diagnostic yield of a biopsy varies widely in the literature between 64–95%, so the risk of the procedure may be incurred without obtaining a definitive tissue diagnosis. If a patient subsequent to SBRT develops a recurrence, a biopsy can be performed on the recurrence, which may present in a more amenable location to biopsy and may be more molecularly insightful in guiding subsequent therapies than would a biopsy at the time of initial diagnosis.

Full table
Existing literature supporting empiric SBRT
There have been several publications examining the clinical outcomes of empiric SBRT (14,26-32) (Table 2). Although there are no specific prospective trials examining empiric versus biopsy-confirmed SBRT, many large prospective studies permitted patients to be enrolled without a biopsy, including the ROSEL trial (14), although most trials required biopsy-proven malignancy, such as in RTOG 0236 (11). As shown in Table 2, inoperable patients comprise 62–88% of the population in these retrospective studies. Toxicity rates, a critical consideration in empiric SBRT, are low, including radiation pneumonitis and rib fractures (<5%) (26,27). In the two studies of 398 and 591 patients who had a PD versus no PD [clinical diagnosis (CD)], no differences in local control (LC), progression-free survival (PFS), or OS were identified, supporting the hypothesis that most of these CD patients who were empirically treated truly did have lung cancers (28,29).
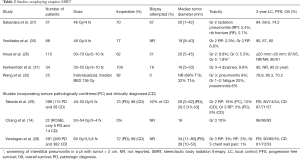
Full table
Tools available for predicting the probability of malignancy
To quantify a given patient’s pretest probability of malignancy, multiple tools have been studied. In the screened population, a calculator was developed from the Pan-Canadian Early Detection of Lung Cancer Study and validated through the British Columbia Cancer Agency. Predictors of malignancy included older age, female sex, family history of lung cancer, emphysema, larger nodule size, location of the nodule in the upper lobe, part-solid nodule type, lower nodule count, and lesion spiculations. Their model had an impressive area under the receiver operating characteristic curve (AUC) of 0.94, even for small nodules (33).
There have also been prediction models developed in the non-screened population, where the prevalence of lung cancer diagnosed after imaging is higher (34). Investigators from the Mayo Clinic examined patients with non-calcified nodules between 4 and 30 mm in diameter, with risk calculated for age (OR 1.04), smoking history (2.2), extra-thoracic cancer <5 years before nodule detection (3.8), nodule diameter (1.14/mm), spiculations (2.8), and upper lobe location (2.2) (35,36). While positron emission tomography (PET) imaging can have clear benefits in diagnosis, staging, prognostication, and target volume delineation in lung cancer (37-39), Herder et al. performed an external validation model of this Swensen analysis on the added value of 18-FDG PET and did not find the AUC was significantly different from the model without PET (40). In summation, studies have shown that these models are similar in accuracy to clinical judgment (41), but overall physicians tend to somewhat overestimate the risk of malignancy.
Determining the appropriate pretest probability threshold for empiric SBRT
The pretest probability threshold above which it is acceptable to perform a therapeutic procedure in this setting, either surgery or SBRT, is not well-defined. The American College of Chest Physicians (ACCP) Evidence-based Clinical Practice Guidelines states that it is reasonable to consider SBRT above a pretest probability of 65% (34); however, other organizations have suggested a higher threshold, including the International Association for the Study of Lung Cancer (IASLC) suggesting that no more than 15% of surgical specimens resulting from a positive screened population should result in benign findings (8) (Table 3). Despite this recommendation, 25% of procedures performed in the NLST had benign findings (4), and the positive predictive value was only 3.8% for 4 mm or larger nodules. These threshold differences in pre-test probability previously recommended between surgical and SBRT populations may, in part, be based on differences in toxicity profiles between surgery and SBRT.

Full table
The question of an appropriate threshold has also been examined specifically within the context of SBRT. Louie et al. constructed a decision tree and Markov model to evaluate the relative merits of surveillance, a PET-directed SBRT strategy (without pathology), or a PET-biopsy-SBRT strategy. They found that the threshold between the two strategies was 85%, which they confirmed on probabilistic sensitivity analysis. The threshold was most sensitive to the diagnostic sensitivity of transthoracic biopsy and the detection rate of false negatives on CT surveillance (42). This is in keeping with the threshold of IASLC and that has been proposed by other investigators (43).
Understanding the combination of clinical and radiographic findings recommended before performing empiric SBRT
As is detailed below, appropriate patient selection for SBRT should be based on a combination of clinical and radiographic findings, including interval lesion growth, size of the suspected malignancy, radiographic appearance of the nodule, patient smoking and prior lung cancer history, and FDG avidity on PET/CT.
Staging procedures recommended for patients undergoing empiric SBRT
In addition to CT chest and PET imaging, NCCN guidelines recommend MRI brain for all patients with a ≥3 cm lesion. A recent institutional report identified a 2.4% risk of asymptomatic brain metastases in tumors <3 cm and 5.6% in tumors up to 5 cm who were radiologically node negative (44). None of the stage II patients were found to have brain metastases, whereas 6.1% of stage IIIA had asymptomatic brain disease. As such, while we recommend that all empiric SBRT patients with lesions >3 cm undergo MRI brain, the requirement could potentially be relaxed for PET-CT and pathologically confirmed node negative patients with tumors >3 cm but ≤5 cm. Whenever feasible, we recommend nodal staging with EBUS for all lesions, acknowledging that it may be acceptable to forego this procedure in patients with no evidence of radiographically enlarged or metabolically active lymph nodes on diagnostic CT chest and PET/CT, respectively, and especially in those with <2 cm lesions and peripherally located lesions (34,45,46). Patients should also undergo pulmonary function testing to better assess their risk of pulmonary complications from treatment and basic laboratory studies to evaluate for an active infection that could be presenting falsely on imaging as a clinical lung cancer.
Considerations for empiric SBRT for suspected tumors >3 cm
If a suspected tumor is >3 cm and identified on either a screening or non-screening scan, it is considered a mass and is malignant until proven otherwise, although still it is important to recognize that that many lesions >3 cm are benign. PET/CT should be obtained. If the tumor is FDG-avid, a biopsy cannot be safely obtained, and there is no clinical suspicion for a benign process, we suggest proceeding with empiric SBRT. If the mass is not FDG-avid, it is much less likely to be malignant. In such cases, we recommend further workup for a benign process and, if no diagnosis is obtained, short-interval serial imaging. As SBRT is increasingly being considered in tumors >5 cm, these same considerations can be applied (47,48). However, as the risk of nodal failures is particularly high in patients with large primary tumors with radiographically negative nodal disease (49), nodal sampling with primary tumor biopsy should be considered whenever clinically feasible.
Empiric SBRT for solitary pulmonary nodules ≤3 cm
There are multiple approaches to consider in the evaluation of patients with solitary pulmonary nodules ≤3 cm. First, we can consider the AACP division of nodules. They separate their management recommendations into solid nodules >8 and ≤8 mm. For nodules >8 mm, if the risk of malignancy is 5–65%, they recommend PET/CT be performed. If the risk if >65%, they recommend against PET/CT and instead recommend a surgical diagnosis. Surgical diagnosis should also be pursued if the nodule is FDG-avid on PET or positive by another functional imaging test and when a fully informed patient prefers a definitive diagnostic procedure. As shown in Table 3, many advocate instead for a clinical probability threshold of >85%, and we agree this is a good threshold. If the pretest probability using the Mayo Clinic calculator is >85%, then it is likely acceptable to proceed with empiric SBRT (32). If the pretest probability is between 65% and 85%, PET/CT is a reasonable approach, and, if FDG-avid to proceed with SBRT. If the nodule is not FDG-avid, patients can undergo short-interval CT at 3 months, and clinicians should use the volume doubling time (VDT) guidelines as described below.
An alternative approach is to use the more recently developed Lung-RAD® system (Table 4), which was created by the American College of Radiology in 2014 as a lexicon to describe lung nodules on screening (50). They increased the size of a positive non-solid (ground-glass) nodule to 2 cm from 6 mm, and the nodule can be considered benign if stable over 3 months (versus 2 years in the NLST). Lung-RAD® has been shown to increase the positive predictive value in CT-screened patients by a factor of 2.5, to 17.3%, without increasing the number of false-negative results (51). Patients who fall into Lung-RAD® category 4A or 4B can undergo PET/CT.

Full table
If the nodule is FDG-avid (SUV ≥2), then empiric SBRT is a reasonable approach. If it is not FDG-avid, then serial observation is likely reasonable. Based on the NELSON CT-screening study, the lesion was considered positive if it had growth on serial scans (defined in this study as 25% growth by volume) and if the VDT was <400 days, or if a new solid component had emerged in a previously non-solid nodule (52). Therefore, if the projected VDT is <400 days based on short-interval scan, the argument for empiric SBRT is further strengthened. In summary, if the pretest probability is >85% based on any calculator, then empiric SBRT is an acceptable option.
Empiric SBRT for ground glass nodules
For pure ground glass nodules without a solid component, the AACP recommends annual surveillance for nodules >5 mm. For larger, part-solid but >50% ground glass nodules, lesions should be managed as solid nodules >8 mm (34), and thus managed according to the discussion above.
Conclusions
We have attempted to apply rationalism to empiricism; we have discussed the strongest evidence currently available and clinical considerations regarding the often-encountered and challenging situation of empiric SBRT for patients with peripheral clinical stage I NSCLC, where biopsy confirmation is too high-risk from a pulmonary function perspective or unable to be obtained safely for other medical or accessibility reasons. We have discussed the scope of the problem and the risks and benefits of empiric SBRT, and we defined the appropriate pretest probability threshold and tools available for predicting this threshold. We provide considerations and recommendations for the clinical and radiographic findings to warrant empiric SBRT, and we divide our recommendation first by size, taking into account recent data from the screened and non-screened populations.
We endorse the American Society for Radiation Oncology guidelines on SBRT for early stage NSCLC recommending that a biopsy prior to SBRT be obtained whenever possible to confirm a histologic diagnosis of malignancy (53). We also recommend that patients without tissue confirmation of malignancy should be discussed in a multidisciplinary manner to reach a consensus on whether the lesion is radiographically and clinically consistent with a lung cancer. The decision to proceed or not proceed with empiric SBRT must be an individualized and multidisciplinary decision, taking into account patient- and tumor-specific factors as outlined in these guidelines. Future research will help further define the pretest probability and continue to improve outcomes after SBRT (54,55).
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Simone CB 2nd, Wildt B, Haas AR, et al. Stereotactic body radiation therapy for lung cancer. Chest 2013;143:1784-90. [Crossref] [PubMed]
- Simone CB 2nd, Dorsey JF. Additional data in the debate on stage I non-small cell lung cancer: surgery versus stereotactic ablative radiotherapy. Ann Transl Med 2015;3:172. [PubMed]
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin 2018;68:7-30. [Crossref] [PubMed]
- Aberle DR, DeMello S, Berg CD, et al. Results of the two incidence screenings in the National Lung Screening Trial. N Engl J Med 2013;369:920-31. [Crossref] [PubMed]
- Humphrey LL, Deffebach M, Pappas M, et al. Screening for Lung Cancer With Low-Dose Computed Tomography: A Systematic Review to Update the U.S. Preventive Services Task Force Recommendation. Ann Intern Med 2013;159:411-20. [Crossref] [PubMed]
- Grills IS, Hope AJ, Guckenberger M, et al. A collaborative analysis of stereotactic lung radiotherapy outcomes for early-stage non-small-cell lung cancer using daily online cone-beam computed tomography image-guided radiotherapy. J Thorac Oncol 2012;7:1382-93. [Crossref] [PubMed]
- McMahon PM, Kong CY, Bouzan C, et al. Cost-effectiveness of computed tomography screening for lung cancer in the United States. J Thorac Oncol 2011;6:1841-8. [Crossref] [PubMed]
- Field JK, Smith RA, Aberle DR, et al. International Association for the Study of Lung Cancer Computed Tomography Screening Workshop 2011 report. J Thorac Oncol 2012;7:10-9. [Crossref] [PubMed]
- Louie AV, Rodrigues GB, Palma DA, et al. Measuring the population impact of introducing stereotactic ablative radiotherapy for stage I non-small cell lung cancer in Canada. Oncologist 2014;19:880-5. [Crossref] [PubMed]
- Verstegen NE, Oosterhuis JWA, Palma DA, et al. Stage I-II non-small-cell lung cancer treated using either stereotactic ablative radiotherapy (SABR) or lobectomy by video-assisted thoracoscopic surgery (VATS): outcomes of a propensity score-matched analysis. Ann Oncol 2013;24:1543-8. [Crossref] [PubMed]
- Timmerman RD, Hu C, Michalski JM, et al. Long-term Results of Stereotactic Body Radiation Therapy in Medically Inoperable Stage I Non-Small Cell Lung Cancer. JAMA Oncol 2018;4:1287-8. [Crossref] [PubMed]
- Schonewolf CA, Heskel M, Doucette A, et al. Five-year Long-term Outcomes of Stereotactic Body Radiation Therapy for Operable Versus Medically Inoperable Stage I Non-small-cell Lung Cancer: Analysis by Operability, Fractionation Regimen, Tumor Size, and Tumor Location. Clin Lung Cancer 2019;20:e63-71. [Crossref] [PubMed]
- Haasbeek CJA, Palma D, Visser O, et al. Early-stage lung cancer in elderly patients: a population-based study of changes in treatment patterns and survival in the Netherlands. Ann Oncol 2012;23:2743-7. [Crossref] [PubMed]
- Chang JY, Senan S, Paul MA, et al. Stereotactic ablative radiotherapy versus lobectomy for operable stage I non-small-cell lung cancer: a pooled analysis of two randomised trials. Lancet Oncol 2015;16:630-7. [Crossref] [PubMed]
- Timmerman RD, Paulus R, Pass HI, et al. Stereotactic Body Radiation Therapy for Operable Early-Stage Lung Cancer: Findings From the NRG Oncology RTOG 0618 Trial. JAMA Oncol 2018;4:1263-6. [Crossref] [PubMed]
- Guckenberger M, Allgäuer M, Appold S, et al. Safety and efficacy of stereotactic body radiotherapy for stage 1 non-small-cell lung cancer in routine clinical practice: a patterns-of-care and outcome analysis. J Thorac Oncol 2013;8:1050-8. [Crossref] [PubMed]
- Valdes G, Solberg T, Heskel M, et al. Using machine learning to predict radiation pneumonitis in patients with stage I non-small cell lung cancer treated with stereotactic body radiation therapy. Phys Med Biol 2016;61:6105-20. [Crossref] [PubMed]
- Chao HH, Valdes G, Luna JM, et al. Exploratory analysis using machine learning to predict for chest wall pain in patients with stage I non-small-cell lung cancer treated with stereotactic body radiation therapy. J Appl Clin Med Phys 2018;19:539-46. [Crossref] [PubMed]
- Timmerman R, McGarry R, Yiannoutsos C, et al. Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J Clin Oncol 2006;24:4833-9. [Crossref] [PubMed]
- Tekatli H, Haasbeek N, Dahele M, et al. Outcomes of Hypofractionated High-Dose Radiotherapy in Poor-Risk Patients with "Ultracentral" Non-Small Cell Lung Cancer. J Thorac Oncol 2016;11:1081-9. [Crossref] [PubMed]
- Bradley JD, Moughan J, Graham MV, et al. A phase I/II radiation dose escalation study with concurrent chemotherapy for patients with inoperable stages I to III non-small-cell lung cancer: phase I results of RTOG 0117. Int J Radiat Oncol Biol Phys 2010;77:367-72. [Crossref] [PubMed]
- Baine MJ, Verma V, Schonewolf CA, et al. Histology Significantly Effects Recurrence and Survival Following SBRT for Early Stage Non-Small Cell Lung Cancer. Lung Cancer 2018;118:20-6. [Crossref] [PubMed]
- Verma V, Simone CB 2nd, Allen PK, et al. Multi-Institutional Experience of Stereotactic Ablative Radiation Therapy for Stage I Small Cell Lung Cancer. Int J Radiat Oncol Biol Phys 2017;97:362-71. [Crossref] [PubMed]
- Verma V, Simone CB 2nd, Allen PK, et al. Outcomes of Stereotactic Body Radiotherapy for T1-T2N0 Small Cell Carcinoma According to Addition of Chemotherapy and Prophylactic Cranial Irradiation: A Multicenter Analysis. Clin Lung Cancer 2017;18:675-81.e1. [Crossref] [PubMed]
- Chowdhry VK, Chowdhry AK, Goldman N, et al. Complications from computed tomography-guided core needle biopsy for patients receiving stereotactic body radiation therapy for early-stage lesions of the lung. Clin Lung Cancer 2014;15:302-6. [Crossref] [PubMed]
- Inoue T, Shimizu S, Onimaru R, et al. Clinical outcomes of stereotactic body radiotherapy for small lung lesions clinically diagnosed as primary lung cancer on radiologic examination. Int J Radiat Oncol Biol Phys 2009;75:683-7. [Crossref] [PubMed]
- Sakanaka K, Matsuo Y, Nagata Y, et al. Safety and effectiveness of stereotactic body radiotherapy for a clinically diagnosed primary stage I lung cancer without pathological confirmation. Int J Clin Oncol 2014;19:814-21. [Crossref] [PubMed]
- Verstegen NE, Lagerwaard FJ, Haasbeek CJA, et al. Outcomes of stereotactic ablative radiotherapy following a clinical diagnosis of stage I NSCLC: comparison with a contemporaneous cohort with pathologically proven disease. Radiother Oncol 2011;101:250-4. [Crossref] [PubMed]
- Takeda A, Kunieda E, Sanuki N, et al. Stereotactic body radiotherapy (SBRT) for solitary pulmonary nodules clinically diagnosed as lung cancer with no pathological confirmation: comparison with non-small-cell lung cancer. Lung Cancer 2012;77:77-82. [Crossref] [PubMed]
- Yoshitake T, Nakamura K, Shioyama Y, et al. Stereotactic body radiation therapy for primary lung cancers clinically diagnosed without pathological confirmation: a single-institution experience. Int J Clin Oncol 2015;20:53-8. [Crossref] [PubMed]
- Harkenrider MM, Bertke MH, Dunlap NE. Stereotactic body radiation therapy for unbiopsied early-stage lung cancer: a multi-institutional analysis. Am J Clin Oncol 2014;37:337-42. [Crossref] [PubMed]
- Wang Z, Li AM, Gao J, et al. written on behalf of the AME Lung Cancer Collaborative Group. Clinical outcomes of CyberKnife stereotactic radiosurgery for elderly patients with presumed primary stage I lung cancer. Transl Lung Cancer Res 2017;6:6-13. [Crossref] [PubMed]
- McWilliams A, Tammemagi MC, Mayo JR, et al. Probability of cancer in pulmonary nodules detected on first screening CT. N Engl J Med 2013;369:910-9. [Crossref] [PubMed]
- Gould MK, Donington J, Lynch WR, et al. Evaluation of individuals with pulmonary nodules: when is it lung cancer? Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 2013;143:e93S-120S.
- Swensen SJ, Silverstein MD, Ilstrup DM, et al. The probability of malignancy in solitary pulmonary nodules. Application to small radiologically indeterminate nodules. Arch Intern Med 1997;157:849-55. [Crossref] [PubMed]
- Swensen SJ, Silverstein MD, Edell ES, et al. Solitary pulmonary nodules: clinical prediction model versus physicians. Mayo Clin Proc 1999;74:319-29. [Crossref] [PubMed]
- Salavati A, Duan F, Snyder BS, et al. Optimal FDG PET/CT volumetric parameters for risk stratification in patients with locally advanced non-small cell lung cancer: results from the ACRIN 6668/RTOG 0235 trial. Eur J Nucl Med Mol Imaging 2017;44:1969-83. [Crossref] [PubMed]
- Simone CB 2nd, Houshmand S, Kalbasi A, et al. PET-based thoracic radiation oncology. PET Clin 2016;11:319-32. [Crossref] [PubMed]
- Verma V, Choi JI, Sawant A, et al. Use of PET and Other Functional Imaging to Guide Target Delineation in Radiation Oncology. Semin Radiat Oncol 2018;28:171-7. [Crossref] [PubMed]
- Herder GJ, van Tinteren H, Golding RP, et al. Clinical prediction model to characterize pulmonary nodules: validation and added value of 18F-fluorodeoxyglucose positron emission tomography. Chest 2005;128:2490-6. [Crossref] [PubMed]
- Balekian AA, Silvestri GA, Simkovich SM, et al. Accuracy of clinicians and models for estimating the probability that a pulmonary nodule is malignant. Ann Am Thorac Soc 2013;10:629-35. [Crossref] [PubMed]
- Louie AV, Senan S, Patel P, et al. When is a biopsy-proven diagnosis necessary before stereotactic ablative radiotherapy for lung cancer? A decision analysis. Chest 2014;146:1021-8. [Crossref] [PubMed]
- Senan S, Paul MA, Lagerwaard FJ. Treatment of early-stage lung cancer detected by screening: surgery or stereotactic ablative radiotherapy? Lancet Oncol 2013;14:e270-4. [Crossref] [PubMed]
- Rice SR, Molitoris JK, Vyfhuis MAL, et al. Lymph Node Size Predicts for Asymptomatic Brain Metastases in Patients With Non-small-cell Lung Cancer at Diagnosis. Clin Lung Cancer 2019;20:e107-14. [Crossref] [PubMed]
- Schonewolf CA, Verma V, Post CM, et al. Outcomes of invasive mediastinal nodal staging versus positron emission tomography staging alone for early-stage non-small cell lung cancer treated with stereotactic body radiation therapy. Lung Cancer 2018;117:53-9. [Crossref] [PubMed]
- Verma V, Schonewolf CA, Cushman TR, et al. Impact of Enlarged Nonhypermetabolic Lymph Nodes on Outcomes After Stereotactic Body Radiotherapy for Early-Stage Non-Small-Cell Lung Cancer. Clin Lung Cancer 2018;19:502-10. [Crossref] [PubMed]
- Verma V, Shostrom VK, Zhen W, et al. Influence of Fractionation Scheme and Tumor Location on Toxicities After Stereotactic Body Radiation Therapy for Large (≥5 cm) Non-Small Cell Lung Cancer: A Multi-institutional Analysis. Int J Radiat Oncol Biol Phys 2017;97:778-85. [Crossref] [PubMed]
- Verma V, McMillian MT, Grover S, et al. Stereotactic body radiation therapy and the influence of chemotherapy on overall survival for large (≥5 centimeter) non-small cell lung cancer. Int J Radiat Oncol Biol Phys 2017;97:146-54. [Crossref] [PubMed]
- Verma V, Shostrom VK, Kumar SS, et al. Multi-institutional experience of stereotactic body radiotherapy for large (≥5 centimeters) non-small cell lung tumors. Cancer 2017;123:688-96. [Crossref] [PubMed]
- Lung CT Screening Reporting and Data System (Lung-RADS) [computer program] 2014. Available online: https://www.acr.org/Clinical-Resources/Reporting-and-Data-Systems/Lung-Rads
- McKee BJ, Regis SM, McKee AB, et al. Performance of ACR Lung-RADS in a Clinical CT Lung Screening Program. J Am Coll Radiol 2016;13:R25-9. [Crossref] [PubMed]
- van Klaveren RJ, Oudkerk M, Prokop M, et al. Management of lung nodules detected by volume CT scanning. N Engl J Med 2009;361:2221-9. [Crossref] [PubMed]
- Videtic GMM, Donington J, Giuliani M, et al. Stereotactic body radiation therapy for early-stage non-small cell lung cancer: Executive Summary of an ASTRO Evidence-Based Guideline. Pract Radiat Oncol 2017;7:295-301. [Crossref] [PubMed]
- Hasan N, Kumar R, Kavuru MS. Lung cancer screening beyond low-dose computed tomography: the role of novel biomarkers. Lung 2014;192:639-48. [Crossref] [PubMed]
- Frick MA, Kao GD, Aguarin L, et al. Circulating Tumor Cell Assessment in Presumed Early Stage Non-Small Cell Lung Cancer Patients Treated with Stereotactic Body Radiation Therapy: A Prospective Pilot Study. Int J Radiat Oncol Biol Phys 2018;102:536-42. [Crossref] [PubMed]


