Beyond ALK-RET, ROS1 and other oncogene fusions in lung cancer
Introduction
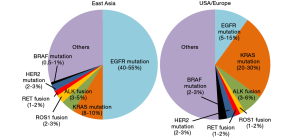
Oncogene activation is a critical step toward the development of non-small cell lung cancer (NSCLC), particularly lung adenocarcinoma (LADC); these activated genes are called driver oncogenes (1-3). Representative driver oncogenes include EGFR, KRAS, BRAF, and HER2/ERBB2, which are activated by missense and/or insertion/deletion mutations, and the ALK gene, which is activated by fusion to other genes (called partner genes) (Figure 1). Aberrations of these genes are mutually exclusively detected in LADC; therefore, they are believed to drive LADC development. Suppressing the activity of aberrant gene products inhibits the growth of LADC cells harboring oncogenic aberrations in the corresponding driver genes. Indeed, tyrosine kinase inhibitors (TKIs) have become the standard drug treatment for advanced cases of LADC harboring EGFR mutations and ALK fusions (1,4,5).
In 2012, two additional oncogenes, RET and ROS1, were added to the list of driver oncogenes that are targetable with existing TKIs (Figure 1A) (1,6-8) and clinical trials investigating the efficacy of such TKIs have been conducted. Furthermore, analysis of lung cancer genome and/or transcriptome has identified other gene fusions, including the NTRK1 (9), NRG1 (10,11), and FGFR1/2/3 fusions (12-14), as novel targetable driver genes in a minor fraction of NSCLC cases. In vitro and in vivo experimental data show that existing TKIs are a promising therapy for lung cancer cases that are positive for these novel oncogenic fusions. Here, we review the oncogenic fusions associated with NSCLC and discuss the issues surrounding personalized therapy.
The RET fusion in LADC
The link between the oncogenic RET fusion and LADC was discovered by several groups (including our own) in 2012. The RET gene was fused to the KIF5B and CCDC6 genes in 1-2% of LADC cases (6-8,15,16); none of these positive cases harbored EGFR, KRAS, BRAF, or HER2/ERBB2 mutations or ALK fusions. The RET fusion is mainly detected in young, female, and/or never/light-smoker patients (6,7,17-19). Also, it occurs in adenocarcinoma but not in squamous and small cell lung cancers (SQLC and SCLC) (2,7). LADCs harboring the RET fusion show well- or moderately-differentiated histological features, similar to those of LADCs harboring EGFR mutations; however, a subset of LADCs harboring the RET fusion show mucinous cribriform features, similar to those of ALK fusion-positive LADCs (6,17-19).
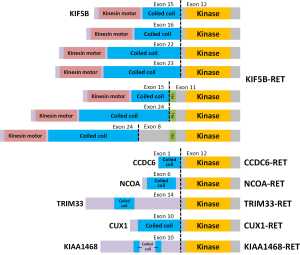
Oncogenic RET variants fused to six partner genes have been identified in lung cancers (10,20,21) (Figure 2). In all of these variants, the coiled-coil domains of the partner proteins induce dimerization of the RET fusion proteins, resulting in constitutive activation of the RET kinase (as in the case of oncogenic ALK fusions). The tumorigenic activity of the RET fusion gene was illustrated by transformation of NIH3T3 cells (6-8) and in a transgenic mouse model in which the KIF5B-RET gene was specifically expressed in lung epithelial cells (22); The tumorigenic activity was suppressed by RET TKIs, indicating its dependence on the kinase activity of the RET protein. Consistent with this, a human LADC cell line derived from a Japanese patient, which carries the CCDC6-RET fusion gene, is sensitive to RET TKIs (23,24). Therefore, LADC cells harboring the RET fusion are in a state of “oncogenic addiction” to constitutive RET kinase activation. This makes the RET fusion a promising therapeutic target.
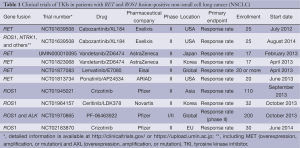
The US Food and Drug Administration (FDA) has approved two multi-kinase inhibitors with RET TKI activity, vandetanib (ZD6474) and cabozantinib (XL184), for the treatment of advanced medullary thyroid cancer in which activating RET mutations are observed in >50% of cases (16). Five phase II clinical trials are currently examining the therapeutic effects of RET TKIs against RET fusion-positive NSCLCs (Table 1). These trials have single-arm open-label designs, with response rate as the primary endpoint. Our own group is conducting one of these phase II clinical trial in Japan (UMIN00001009). This trial, designated “LURET (lung cancer with RET rearrangement study)”, is designed to investigate the therapeutic efficacy of vandetanib against NSCLC. We are using a RT-PCR-based screening method to select patients with RET fusion-positive tumors. This process is being carried out in >170 hospitals via a consortium called “LC-SCRUM (lung cancer genomic screening project for individualized medicine in Japan)”, and >1,000 patients with advanced NSCLC without EGFR mutations have been screened as of Aug 31, 2014 (2). A trial conducted at Memorial Sloan-Kettering Cancer Center (NCT01639508) reported promising responses in the first three patients treated with cabozantinib (20). In addition, another study reported that one patient with LADC harboring a KIF5B-RET fusion showed a positive response to vandetanib (25). Although the number of patients in these studies is small and follow-up is limited, the results provide early proof-of-principle that the RET fusion is targetable by existing TKIs.
Full table
The ROS1 fusion in LADC
The oncogenic ROS1 fusion is present in 1-2% of LADC cases (6), and is likely to be specific for adenocarcinoma (26). The ROS1 gene fuses to several partner genes, although CD74 is the most common (Figure 3) (27-29). As is the case for the RET fusion, the ROS1 fusion occurs in a manner that is mutually exclusive with other known driver oncogene mutations and fusions. The ROS1 fusion is preferentially detected in young, female, and/or never/light-smoker patients (6,18,30-32). LADCs harboring the ROS1 fusion often show mucinous cribriform features (6,18,30,31), similar to those of ALK fusion-positive LADCs. The ROS1 fusion is also likely to be specific for LADC (6,18,30,32).

The transforming activity of the ROS1 fusion gene has been demonstrated in vitro using NIH3T3 cells (6,33) and in vivo using a transgenic mouse model in which the EZR-ROS1 gene is specifically expressed in lung epithelial cells (33). Crizotinib, a TKI approved by the FDA for ALK fusion-positive lung cancer, also inhibits the ROS1 protein due to the structural similarity of the kinase domains of ROS1 and ALK proteins. In fact, the LADC cell line, HCC78, which harbors a SLC34A2-ROS1 fusion, is sensitive to crizotinib (26,32). Thus, LADC cells harboring the ROS1 fusion are in a state of “oncogenic addiction” to constitutive ROS1 kinase activation. In contrast to the RET and ALK fusions, constitutive activation of the ROS1 kinase protein is unlikely due to dimerization of ROS1 fusion proteins since the majority of ROS1 partner proteins lack dimerization domains (27) (Figure 3).
A phase I trial (NCT00585195) examining the efficacy of crizotinib against ROS1 fusion-positive NSCLC showed an objective response rate of 60% (27). Other studies (32,34-36) report that patients with LADC harboring a ROS1 fusion show a near-complete or partial response to crizotinib. Therefore, molecular-targeted therapy using crizotinib (and other ROS1 TKIs) appears promising. Five phase II or I/II clinical trials have been conducted to examine the therapeutic effects of ROS1 TKIs against ROS1 fusion-positive NSCLCs (Table 1). The LC-SCRUM consortium is currently screening ROS1 fusion-positive tumors in Japan and ROS1 fusion-positive patients are being enrolled in a crizotinib trial (NCT01945021).
Other protein kinase fusions in LADC
Other oncogenic fusions of protein kinase genes have been detected in LADCs that are negative for known driver oncogene aberrations (Figure 4A). Oncogenic fusions of the NTRK1 gene (which encodes a nerve growth factor receptor, TRKA) with the CD74 and MPRIP genes were recently identified in 3% of patients within an American cohort (9). However, other LADC cohorts, including a TCGA-USA cohort (n=230), a Korean cohort (n=87), and our own NCC-Japan cohort (n=200, unpublished data), contained no NTRK1 fusion-positive cases (9). Thus, the prevalence of NTRK1 fusion remains unclear. A few TKIs (ARRY-470, CEP-701, and crizotinib) that suppress the activity of the TRKA protein kinase also suppress the NIH3T3-transforming activity of the NTRK fusion gene (9). Notably, a LADC patient harboring the MPRIP-NTRK1 fusion showed a minor therapeutic response to crizotinib (9). An ongoing clinical trial (NCT01639508) includes not only patients positive for the RET and ROS1 fusions, but also patients positive for the NTRK1 fusion (Table 1).

The AXL-MBIP and SCAF11-PDGFRA fusions, two more protein kinase gene fusions (Figure 4A), were each detected in a single case of LADC in a Korean cohort of 200 patients (29). Since these fusions were not detected in either the TCGA-USA cohort (n=230) (28) or our own NCC-Japan cohort (n=200, unpublished data), they may only occur in a very small subset of LADC cases.
Multiple oncogenic fusions in invasive mucinous LADC
Invasive mucinous adenocarcinoma (IMA) of the lungs, which accounts for 2-10% of all LADC cases in Japan, the USA, and Europe, is thought to be a distinct histological type of LADC that commonly (>50%) harbors KRAS mutations (37,38). We recently identified multiple oncogene fusions involving the NRG1 (neuregulin), ERBB4, BRAF, ALK, and RET genes as drivers for the development of IMA in the absence of KRAS mutations (10) (Figure 4B). Among these, the CD74-NRG1 fusion was the most common (5-15%). The CD74-NRG1 fusion has also been detected in another Japanese IMA cohort and in a Taiwanese IMA cohort (11,39). The fusion product acts as a ligand for HER2:HER3 and causes anchorage-independent growth of NIH3T3 cells (9,10,11). Its transforming activity is suppressed by HER2 inhibitors that are approved for clinical use, including lapatinib and afatinib (10), suggesting that IMAs may be amenable to personalized therapy.
FGFR1/2/3 fusions in SQLC
Amplification of the FGFR1 gene has been identified as a major oncogene aberration in approximately 10% of SQLC cases (40), whereas activating mutations in FGFR1, FGFR2, and FGFR3 are detected in a small subset of SQLC cases (41). Recent studies have detected fusions of the FGFR1, FGFR2, and FGFR3 genes to several partner genes in SQLC (Figure 4C) (13,14,28). In particular, the FGFR3-TACC3 fusion, which is detected in 3% of glioblastoma multiforme cases (42), was recurrently observed in a 2-3% of LSQC cases. The FGFR3-TACC3 fusion gene induces cell transformation and accelerated growth. Both cell growth and tumorigenicity are suppressed by FGFR TKIs (13). Importantly, several clinical trials examining the efficacy of FGFR TKIs against SQLC harboring mutation/amplification of the FGFR genes are ongoing, although broadening the inclusion criteria for such clinical trials would be beneficial.
Diagnosis of fusion-positive cases
The findings discussed to date provide a strong rationale for developing precision medicine approaches based on targeting oncogene fusions in LADC and LSQC. Since this form of therapy is applicable only to a subset of LADC and LSQC cases, it is important that we develop suitable diagnostic methods that are able to identify fusion-positive cases (43). The diagnosis of ALK fusion-positive lung cancer is based on fluorescence in situ hybridization (FISH) either with or without immunohistochemistry (IHC) (44). FISH and IHC are also suitable for the diagnosis of ROS1 fusion (45,46); however, IHC is not suitable for the diagnosis of RET fusion (7,8,19).
Because only very small amounts of material can be obtained from biopsies, there is a need to develop diagnostic systems that enable simultaneous examination of multiple gene fusions in routine formalin-fixed and paraffin-embedded (FFPE) clinical specimens. However, because the FFPE technique damages DNA, the robustness against DNA qualities is needed for the diagnostic systems. In addition, most of the samples that are subjected to testing are small biopsies; therefore, the system must also be able to deal with limited amounts of tissue and/or extracted DNA/RNA. Accurate and sensitive profiling must be achieved, even when the proportion of tumor cells within the specimens is low.
Representative systems are currently being developed that will enable multiple, robust, and sensitive diagnoses (Table 2). Some employ the method of target re-sequencing of tens to hundreds of genes using DNA or RNA extracted from tumor tissues (47,48), while others employ quantitative RT-PCR or RNA molecule counting (21,49,50). Optimizing these (or other equivalent) systems for use in the clinic will greatly facilitate the progress toward precision medicine for lung cancer.

Full table
Perspective: issues still to be investigated
In vitro/in vivo experiments and the responses of the few patients examined in trials suggest that the therapies described in this review hold promise. However, innate and acquired resistance to TKIs may become a problem, as is the case for TKIs targeting the ALK and EGFR proteins. The mechanisms underlying resistance are beginning to be unraveled and several next-generation TKIs have been developed to treat resistant ALK fusion and EGFR mutations (5,51). This is good news because some ROS1 fusion-positive cases also have acquired resistance to crizotinib (52). Further studies should be done on the resistance of other fusions to TKIs so that lung cancers harboring novel fusions can be treated effectively.
Preventing the development of lung cancer via oncogenic fusions is another issue to be tackled by those involved in lung cancer medicine. LADCs harboring oncogene fusions are mainly observed in never/light smokers; therefore, preventive methods other than smoking cessation are necessary. We have been investigating the molecular mechanisms underlying chromosome inversions that generate oncogenic RET fusions in LADC by cloning genomic segments that contain breakpoint junctions (53). We found that inversions were most likely caused by the mis-repair of DNA strand breaks, which occurred in a region spanning a few Kb within the RET gene (the region in which DNA strand breaks leading to RET rearrangements in papillary thyroid tumors also frequently occur) (53). Thus, tobacco-independent DNA strand breaks are likely to trigger development of the RET fusion. To the best of our knowledge, no studies have elucidated the structure of the breakpoints in ALK, ROS1, and other fusions. Further examination of the molecular processes underlying gene fusion, as well as identifying the endogenous/exogenous factors that cause DNA breaks, will provide the key to preventing the development of lung cancers harboring oncogenic gene fusions.
Acknowledgements
The authors thank all the collaborators in the NCC and LC-SCRUM/LURET studies.
Funding: This work was supported in part by Grants-in-Aid from the Ministry of Health, Labor, and Welfare.
Disclosure: The authors declare no conflict of interest.
References
- Oxnard GR, Binder A, Jänne PA. New targetable oncogenes in non-small-cell lung cancer. J Clin Oncol 2013;31:1097-104. [PubMed]
- Kohno T, Tsuta K, Tsuchihara K, et al. RET fusion gene: translation to personalized lung cancer therapy. Cancer Sci 2013;104:1396-400. [PubMed]
- Pao W, Hutchinson KE. Chipping away at the lung cancer genome. Nat Med 2012;18:349-51. [PubMed]
- Li T, Kung HJ, Mack PC, et al. Genotyping and genomic profiling of non-small-cell lung cancer: implications for current and future therapies. J Clin Oncol 2013;31:1039-49. [PubMed]
- Shaw AT, Engelman JA. ALK in lung cancer: past, present, and future. J Clin Oncol 2013;31:1105-11. [PubMed]
- Takeuchi K, Soda M, Togashi Y, et al. RET, ROS1 and ALK fusions in lung cancer. Nat Med 2012;18:378-81. [PubMed]
- Kohno T, Ichikawa H, Totoki Y, et al. KIF5B-RET fusions in lung adenocarcinoma. Nat Med 2012;18:375-7. [PubMed]
- Lipson D, Capelletti M, Yelensky R, et al. Identification of new ALK and RET gene fusions from colorectal and lung cancer biopsies. Nat Med 2012;18:382-4. [PubMed]
- Vaishnavi A, Capelletti M, Le AT, et al. Oncogenic and drug-sensitive NTRK1 rearrangements in lung cancer. Nat Med 2013;19:1469-72. [PubMed]
- Nakaoku T, Tsuta K, Ichikawa H, et al. Druggable oncogene fusions in invasive mucinous lung adenocarcinoma. Clin Cancer Res 2014;20:3087-93. [PubMed]
- Fernandez-Cuesta L, Plenker D, Osada H, et al. CD74-NRG1 Fusions in Lung Adenocarcinoma. Cancer Discov 2014;4:415-22. [PubMed]
- Wang R, Wang L, Li Y, et al. FGFR1/3 tyrosine kinase fusions define a unique molecular subtype of non-small cell lung cancer. Clin Cancer Res 2014;20:4107-14. [PubMed]
- Wu YM, Su F, Kalyana-Sundaram S, et al. Identification of targetable FGFR gene fusions in diverse cancers. Cancer Discov 2013;3:636-47. [PubMed]
- Majewski IJ, Mittempergher L, Davidson NM, et al. Identification of recurrent FGFR3 fusion genes in lung cancer through kinome-centred RNA sequencing. J Pathol 2013;230:270-6. [PubMed]
- Ju YS, Lee WC, Shin JY, et al. A transforming KIF5B and RET gene fusion in lung adenocarcinoma revealed from whole-genome and transcriptome sequencing. Genome Res 2012;22:436-45. [PubMed]
- Mulligan LM. RET revisited: expanding the oncogenic portfolio. Nat Rev Cancer 2014;14:173-86. [PubMed]
- Tsuta K, Kohno T, Yoshida A, et al. RET-rearranged non-small-cell lung carcinoma: a clinicopathological and molecular analysis. Br J Cancer 2014;110:1571-8. [PubMed]
- Pan Y, Zhang Y, Li Y, et al. ALK, ROS1 and RET fusions in 1139 lung adenocarcinomas: a comprehensive study of common and fusion pattern-specific clinicopathologic, histologic and cytologic features. Lung Cancer 2014;84:121-6. [PubMed]
- Wang R, Hu H, Pan Y, et al. RET fusions define a unique molecular and clinicopathologic subtype of non-small-cell lung cancer. J Clin Oncol 2012;30:4352-9. [PubMed]
- Drilon A, Wang L, Hasanovic A, et al. Response to Cabozantinib in patients with RET fusion-positive lung adenocarcinomas. Cancer Discov 2013;3:630-5. [PubMed]
- Lira ME, Choi YL, Lim SM, et al. A single-tube multiplexed assay for detecting ALK, ROS1, and RET fusions in lung cancer. J Mol Diagn 2014;16:229-43. [PubMed]
- Saito M, Ishigame T, Tsuta K, et al. A mouse model of KIF5B-RET fusion-dependent lung tumorigenesis. Carcinogenesis 2014;35:2452-6. [PubMed]
- Suzuki M, Makinoshima H, Matsumoto S, et al. Identification of a lung adenocarcinoma cell line with CCDC6-RET fusion gene and the effect of RET inhibitors in vitro and in vivo. Cancer Sci 2013;104:896-903. [PubMed]
- Matsubara D, Kanai Y, Ishikawa S, et al. Identification of CCDC6-RET fusion in the human lung adenocarcinoma cell line, LC-2/ad. J Thorac Oncol 2012;7:1872-6. [PubMed]
- Gautschi O, Zander T, Keller FA, et al. A patient with lung adenocarcinoma and RET fusion treated with vandetanib. J Thorac Oncol 2013;8:e43-4. [PubMed]
- Davies KD, Le AT, Theodoro MF, et al. Identifying and targeting ROS1 gene fusions in non-small cell lung cancer. Clin Cancer Res 2012;18:4570-9. [PubMed]
- Davies KD, Doebele RC. Molecular pathways: ROS1 fusion proteins in cancer. Clin Cancer Res 2013;19:4040-5. [PubMed]
- Cancer Genome Atlas Research Network. Comprehensive molecular profiling of lung adenocarcinoma. Nature 2014;511:543-50. [PubMed]
- Seo JS, Ju YS, Lee WC, et al. The transcriptional landscape and mutational profile of lung adenocarcinoma. Genome Res 2012;22:2109-19. [PubMed]
- Yoshida A, Kohno T, Tsuta K, et al. ROS1-rearranged lung cancer: a clinicopathologic and molecular study of 15 surgical cases. Am J Surg Pathol 2013;37:554-62. [PubMed]
- Chen YF, Hsieh MS, Wu SG, et al. Clinical and the prognostic characteristics of lung adenocarcinoma patients with ROS1 fusion in comparison with other driver mutations in East Asian populations. J Thorac Oncol 2014;9:1171-9. [PubMed]
- Bergethon K, Shaw AT, Ou SH, et al. ROS1 rearrangements define a unique molecular class of lung cancers. J Clin Oncol 2012;30:863-70. [PubMed]
- Arai Y, Totoki Y, Takahashi H, et al. Mouse model for ROS1-rearranged lung cancer. PLoS One 2013;8:e56010. [PubMed]
- Bos M, Gardizi M, Schildhaus HU, et al. Complete metabolic response in a patient with repeatedly relapsed non-small cell lung cancer harboring ROS1 gene rearrangement after treatment with crizotinib. Lung Cancer 2013;81:142-3. [PubMed]
- Komiya T, Thomas A, Khozin S, et al. Response to crizotinib in ROS1-rearranged non-small-cell lung cancer. J Clin Oncol. 2012 Sep 20;30:3425-6; author reply 3426. [PubMed]
- Chiari R, Buttitta F, Iacono D, et al. Dramatic Response to Crizotinib in ROS1 Fluorescent In Situ Hybridization- and Immunohistochemistry-Positive Lung Adenocarcinoma: A Case Series. Clin Lung Cancer 2014;15:470-4. [PubMed]
- Tsuta K, Kawago M, Inoue E, et al. The utility of the proposed IASLC/ATS/ERS lung adenocarcinoma subtypes for disease prognosis and correlation of driver gene alterations. Lung Cancer 2013;81:371-6. [PubMed]
- Yoshizawa A, Sumiyoshi S, Sonobe M, et al. Validation of the IASLC/ATS/ERS lung adenocarcinoma classification for prognosis and association with EGFR and KRAS gene mutations: analysis of 440 Japanese patients. J Thorac Oncol 2013;8:52-61. [PubMed]
- Gow CH, Wu SG, Chang YL, et al. Multidriver mutation analysis in pulmonary mucinous adenocarcinoma in Taiwan: identification of a rare CD74-NRG1 translocation case. Med Oncol 2014;31:34. [PubMed]
- Weiss J, Sos ML, Seidel D, et al. Frequent and focal FGFR1 amplification associates with therapeutically tractable FGFR1 dependency in squamous cell lung cancer. Sci Transl Med 2010;2:62ra93. [PubMed]
- Liao RG, Jung J, Tchaicha J, et al. Inhibitor-sensitive FGFR2 and FGFR3 mutations in lung squamous cell carcinoma. Cancer Res 2013;73:5195-205. [PubMed]
- Singh D, Chan JM, Zoppoli P, et al. Transforming fusions of FGFR and TACC genes in human glioblastoma. Science 2012;337:1231-5. [PubMed]
- Ou SH, Soo RA, Kubo A, et al. Will the Requirement by the US FDA to Simultaneously Co-Develop Companion Diagnostics (CDx) Delay the Approval of Receptor Tyrosine Kinase Inhibitors for RTK-Rearranged (ROS1-, RET-, AXL-, PDGFR-α-, NTRK1-) Non-Small Cell Lung Cancer Globally? Front Oncol 2014;4:58. [PubMed]
- Iwama E, Okamoto I, Harada T, et al. Development of anaplastic lymphoma kinase (ALK) inhibitors and molecular diagnosis in ALK rearrangement-positive lung cancer. Onco Targets Ther 2014;7:375-85. [PubMed]
- Yoshida A, Tsuta K, Wakai S, et al. Immunohistochemical detection of ROS1 is useful for identifying ROS1 rearrangements in lung cancers. Mod Pathol 2014;27:711-20. [PubMed]
- Rimkunas VM, Crosby KE, Li D, et al. Analysis of receptor tyrosine kinase ROS1-positive tumors in non-small cell lung cancer: identification of a FIG-ROS1 fusion. Clin Cancer Res 2012;18:4449-57. [PubMed]
- Frampton GM, Fichtenholtz A, Otto GA, et al. Development and validation of a clinical cancer genomic profiling test based on massively parallel DNA sequencing. Nat Biotechnol 2013;31:1023-31. [PubMed]
- Conley BA, Doroshow JH. Molecular analysis for therapy choice: NCI MATCH. Semin Oncol 2014;41:297-9. [PubMed]
- Zhao C, Li X, Li J, et al. Detecting ALK, ROS1 and RET fusion genes in cell block samples. Transl Oncol 2014;7:363-7. [PubMed]
- Zheng Z, Liebers M, Zhelyazkova B, et al. Anchored multiplex PCR for targeted next-generation sequencing. Nat Med 2014;20:1479-84. [PubMed]
- Chong CR, Jänne PA. The quest to overcome resistance to EGFR-targeted therapies in cancer. Nat Med 2013;19:1389-400. [PubMed]
- Awad MM, Katayama R, McTigue M, et al. Acquired resistance to crizotinib from a mutation in CD74-ROS1. N Engl J Med 2013;368:2395-401. [PubMed]
- Mizukami T, Shiraishi K, Shimada Y, et al. Molecular mechanisms underlying oncogenic RET fusion in lung adenocarcinoma. J Thorac Oncol 2014;9:622-30. [PubMed]